

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
null
SMILES: C[C@]12CC[C@H]3[C@@H](CCC4=CC(=O)CC[C@H]34)[C@@H]1CC[C@@]2(O)C#C
InChI Key: InChIKey=VIKNJXKGJWUCNN-XGXHKTLJSA-N
PDB links: 2 PDB IDs match this monomer.
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Progesterone receptor (Homo sapiens (Human)) | BDBM50148732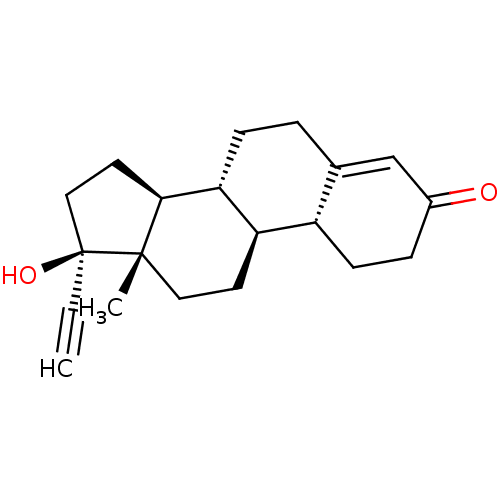 (17-ethynyl-17beta-hydroxyestr-4-en-3-one | 17alpha...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description The binding affinity measured using baculovirus-expressed hPR-A in sf21 cells. | J Med Chem 41: 303-10 (1998) Article DOI: 10.1021/jm9705770 BindingDB Entry DOI: 10.7270/Q2RV0PC8 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Progesterone receptor (Homo sapiens (Human)) | BDBM50148732 (17-ethynyl-17beta-hydroxyestr-4-en-3-one | 17alpha...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB PubMed | n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
R. W. Johnson Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Binding affinity against Progesterone receptor in human TE85 osteosarcoma cells was determined using (Z)-[125I]-17-alpha-(2-iodovinyl)-19-nor-testost... | J Med Chem 38: 4880-4 (1996) BindingDB Entry DOI: 10.7270/Q2KP82SZ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Progesterone receptor (Homo sapiens (Human)) | BDBM50148732 (17-ethynyl-17beta-hydroxyestr-4-en-3-one | 17alpha...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | n/a | n/a | n/a | n/a | 2.20 | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Agonistic activity was measured for modulation of hPR-B (human progesterone receptor) in co-transfected CV-1 cells. | J Med Chem 41: 303-10 (1998) Article DOI: 10.1021/jm9705770 BindingDB Entry DOI: 10.7270/Q2RV0PC8 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Bile salt export pump (Homo sapiens (Human)) | BDBM50148732 (17-ethynyl-17beta-hydroxyestr-4-en-3-one | 17alpha...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.85E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Curated by ChEMBL | Assay Description Ability to inhibit HMG-CoA reductase (HMGR) by cholesterol synthesis inhibition screen (CSI) in rats | Drug Metab Dispos 40: 2332-41 (2012) Article DOI: 10.1124/dmd.112.047068 BindingDB Entry DOI: 10.7270/Q2ZP488M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Rattus norvegicus (Rat)) | BDBM50148732 (17-ethynyl-17beta-hydroxyestr-4-en-3-one | 17alpha...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 120 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Basel Curated by ChEMBL | Assay Description Inhibitory concentration against recombinant rat androgen receptor expressed in Escherichia coli using [3H]methyltrienolone (R 1881) | J Med Chem 48: 5666-74 (2005) Article DOI: 10.1021/jm050403f BindingDB Entry DOI: 10.7270/Q2TM7CBZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Estrogen receptor beta (RAT-Rattus norvegicus) | BDBM50148732 (17-ethynyl-17beta-hydroxyestr-4-en-3-one | 17alpha...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | n/a | 0.630 | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Equilibrium dissociation constant for rat uterine estrogen receptor binding [3H]estradiol | J Med Chem 27: 1131-7 (1984) BindingDB Entry DOI: 10.7270/Q2W95CDM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Bile salt export pump (Homo sapiens (Human)) | BDBM50148732 (17-ethynyl-17beta-hydroxyestr-4-en-3-one | 17alpha...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 4.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc. Curated by ChEMBL | Assay Description Inhibition of human BSEP expressed in fall armyworm sf9 cell plasma membrane vesicles assessed as reduction in vesicle-associated [3H]-taurocholate t... | Toxicol Sci 118: 485-500 (2010) Article DOI: 10.1093/toxsci/kfq269 BindingDB Entry DOI: 10.7270/Q26Q20JN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Progesterone receptor (Homo sapiens (Human)) | BDBM50148732 (17-ethynyl-17beta-hydroxyestr-4-en-3-one | 17alpha...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | n/a | n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Inc. Curated by ChEMBL | Assay Description Dissociation constant for progesterone receptor | J Med Chem 47: 3381-7 (2004) Article DOI: 10.1021/jm030640n BindingDB Entry DOI: 10.7270/Q2KP81NR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Sex hormone-binding globulin (Homo sapiens (Human)) | BDBM50148732 (17-ethynyl-17beta-hydroxyestr-4-en-3-one | 17alpha...) | PDB MMDB NCI pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank Article PubMed | n/a | n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Displacement of [3H]5alpha dihydrotestosterone from human sex hormone binding globulin | J Med Chem 51: 2047-56 (2008) Article DOI: 10.1021/jm7011485 BindingDB Entry DOI: 10.7270/Q2RX9DC2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||