

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM41132 MLS000028542::SMR000058333::TRIAMCINOLONE::cid_31307
SMILES: C[C@]12C[C@H](O)[C@@]3(F)[C@@H](CCC4=CC(=O)C=C[C@]34C)[C@@H]1C[C@@H](O)[C@]2(O)C(=O)CO
InChI Key: InChIKey=GFNANZIMVAIWHM-OBYCQNJPSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Procathepsin L (Homo sapiens (Human)) | BDBM41132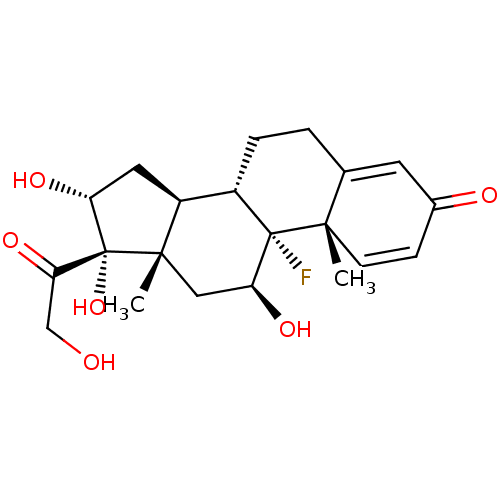 (MLS000028542 | SMR000058333 | TRIAMCINOLONE | cid_...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid UniChem Patents Similars | PCBioAssay | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
PCMD Curated by PubChem BioAssay | Assay Description Screening Center: Penn Center for Molecular Discovery Center Affiliation: University of Pennsylvania Network: Molecular Library Screening Center Netw... | PubChem Bioassay (2008) BindingDB Entry DOI: 10.7270/Q27D2SJC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Multidrug resistance protein 1/Multidrug resistance associated protein 1 (Homo sapiens (Human)) | BDBM41132 (MLS000028542 | SMR000058333 | TRIAMCINOLONE | cid_...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid UniChem Patents Similars | PCBioAssay | n/a | n/a | n/a | n/a | 7.62E+3 | n/a | n/a | n/a | n/a |
SRMLSC Curated by PubChem BioAssay | Assay Description Southern Research Molecular Libraries Screening Center (SRMLSC) Southern Research Institute (Birmingham, Alabama) NIH Molecular Libraries Screening C... | PubChem Bioassay (2007) BindingDB Entry DOI: 10.7270/Q25719FH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Procathepsin L (Homo sapiens (Human)) | BDBM41132 (MLS000028542 | SMR000058333 | TRIAMCINOLONE | cid_...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid UniChem Patents Similars | PCBioAssay | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
PCMD Curated by PubChem BioAssay | Assay Description Screening Center: Penn Center for Molecular Discovery Center Affiliation: University of Pennsylvania Network: Molecular Library Screening Center Netw... | PubChem Bioassay (2009) BindingDB Entry DOI: 10.7270/Q2WH2NDV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2J2 (Homo sapiens (Human)) | BDBM41132 (MLS000028542 | SMR000058333 | TRIAMCINOLONE | cid_...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 9.47E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Tongji University Curated by ChEMBL | Assay Description Inhibition of human recombinant CYP2J2 assessed as reduction in astemizole O-demethylation by LC-MS/MS method | Drug Metab Dispos 41: 60-71 (2012) Article DOI: 10.1124/dmd.112.048264 BindingDB Entry DOI: 10.7270/Q2K0761G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 3A4 (Homo sapiens (Human)) | BDBM41132 (MLS000028542 | SMR000058333 | TRIAMCINOLONE | cid_...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid UniChem Patents Similars | DrugBank Article PubMed | n/a | n/a | 4.91E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tongji University Curated by ChEMBL | Assay Description Inhibition of CYP3A4 in human liver microsomes using testosterone substrate by LC-MS/MS method | Drug Metab Dispos 41: 60-71 (2012) Article DOI: 10.1124/dmd.112.048264 BindingDB Entry DOI: 10.7270/Q2K0761G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2C9 (Homo sapiens (Human)) | BDBM41132 (MLS000028542 | SMR000058333 | TRIAMCINOLONE | cid_...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tongji University Curated by ChEMBL | Assay Description Inhibition of CYP2C9 in human liver microsomes using tolbutamide substrate by LC-MS/MS method | Drug Metab Dispos 41: 60-71 (2012) Article DOI: 10.1124/dmd.112.048264 BindingDB Entry DOI: 10.7270/Q2K0761G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2C19 (Homo sapiens (Human)) | BDBM41132 (MLS000028542 | SMR000058333 | TRIAMCINOLONE | cid_...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tongji University Curated by ChEMBL | Assay Description Inhibition of CYP2C19 in human liver microsomes using omeprazole substrate by LC-MS/MS method | Drug Metab Dispos 41: 60-71 (2012) Article DOI: 10.1124/dmd.112.048264 BindingDB Entry DOI: 10.7270/Q2K0761G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 1A (Homo sapiens (Human)) | BDBM41132 (MLS000028542 | SMR000058333 | TRIAMCINOLONE | cid_...) | PDB MMDB Reactome pathway KEGG B.MOAD DrugBank GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tongji University Curated by ChEMBL | Assay Description Inhibition of CYP1A2 in human liver microsomes using phenacetin substrate by LC-MS/MS method | Drug Metab Dispos 41: 60-71 (2012) Article DOI: 10.1124/dmd.112.048264 BindingDB Entry DOI: 10.7270/Q2K0761G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D6 (Homo sapiens (Human)) | BDBM41132 (MLS000028542 | SMR000058333 | TRIAMCINOLONE | cid_...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tongji University Curated by ChEMBL | Assay Description Inhibition of CYP2D6 in human liver microsomes using bufuralol substrate by LC-MS/MS method | Drug Metab Dispos 41: 60-71 (2012) Article DOI: 10.1124/dmd.112.048264 BindingDB Entry DOI: 10.7270/Q2K0761G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||