

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM10351 1-(4-Methoxybenzyl)-5-(2-(pyridin-3-yl-oxymethyl)-pyrolidine-1-sulfonyl)-1H-indole-2,3-dione::1-[(4-methoxyphenyl)methyl]-5-{[(2S)-2-[(pyridin-3-yloxy)methyl]pyrrolidine-1-]sulfonyl}-2,3-dihydro-1H-indole-2,3-dione::Pyridine Analogue 21c
SMILES: COc1ccc(CN2C(=O)C(=O)c3cc(ccc23)S(=O)(=O)N2CCC[C@H]2COc2cccnc2)cc1
InChI Key: InChIKey=XDHJBIYJRNFDKS-IBGZPJMESA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Caspase-3 (Homo sapiens (Human)) | BDBM10351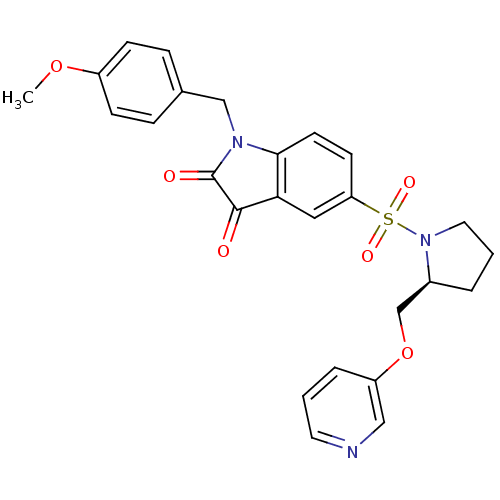 (1-(4-Methoxybenzyl)-5-(2-(pyridin-3-yl-oxymethyl)-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | 4.40 | n/a | 3.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Washington University School of Medicine | Assay Description The substrate peptides terminating in AMC are processed by caspases with or without inhibitors. The amount of AMC released was determined by using a ... | J Med Chem 48: 7637-47 (2005) Article DOI: 10.1021/jm0506625 BindingDB Entry DOI: 10.7270/Q2V40SDN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Caspase-1 (Homo sapiens (Human)) | BDBM10351 (1-(4-Methoxybenzyl)-5-(2-(pyridin-3-yl-oxymethyl)-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Washington University School of Medicine | Assay Description The substrate peptides terminating in AMC are processed by caspases with or without inhibitors. The amount of AMC released was determined by using a ... | J Med Chem 48: 7637-47 (2005) Article DOI: 10.1021/jm0506625 BindingDB Entry DOI: 10.7270/Q2V40SDN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Caspase-3 (Homo sapiens (Human)) | BDBM10351 (1-(4-Methoxybenzyl)-5-(2-(pyridin-3-yl-oxymethyl)-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.89 | n/a | n/a | n/a | n/a | n/a | n/a |
Chugai Pharmaceutical Company Curated by ChEMBL | Assay Description Inhibition of caspase3 | Bioorg Med Chem 20: 5410-5 (2012) Article DOI: 10.1016/j.bmc.2012.03.041 BindingDB Entry DOI: 10.7270/Q2GF0VTJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Caspase-8 (Homo sapiens (Human)) | BDBM10351 (1-(4-Methoxybenzyl)-5-(2-(pyridin-3-yl-oxymethyl)-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Washington University School of Medicine | Assay Description The substrate peptides terminating in AMC are processed by caspases with or without inhibitors. The amount of AMC released was determined by using a ... | J Med Chem 48: 7637-47 (2005) Article DOI: 10.1021/jm0506625 BindingDB Entry DOI: 10.7270/Q2V40SDN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Caspase-3 (Homo sapiens (Human)) | BDBM10351 (1-(4-Methoxybenzyl)-5-(2-(pyridin-3-yl-oxymethyl)-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 450 | n/a | n/a | n/a | n/a |
Washington University School of Medicine Curated by ChEMBL | Assay Description Inhibition of staurosporine induced activation of caspase 3 activation in human Hela cells assessed as hydrolysis of Z-DEVD-R110 substrate by micropl... | Bioorg Med Chem Lett 21: 2192-7 (2011) Article DOI: 10.1016/j.bmcl.2011.03.015 BindingDB Entry DOI: 10.7270/Q2TB176G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Caspase-7 (Homo sapiens (Human)) | BDBM10351 (1-(4-Methoxybenzyl)-5-(2-(pyridin-3-yl-oxymethyl)-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 15.1 | n/a | n/a | n/a | n/a | n/a | n/a |
Washington University School of Medicine | Assay Description The substrate peptides terminating in AMC are processed by caspases with or without inhibitors. The amount of AMC released was determined by using a ... | J Med Chem 48: 7637-47 (2005) Article DOI: 10.1021/jm0506625 BindingDB Entry DOI: 10.7270/Q2V40SDN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Caspase-6 (Homo sapiens (Human)) | BDBM10351 (1-(4-Methoxybenzyl)-5-(2-(pyridin-3-yl-oxymethyl)-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Washington University School of Medicine | Assay Description The substrate peptides terminating in AMC are processed by caspases with or without inhibitors. The amount of AMC released was determined by using a ... | J Med Chem 48: 7637-47 (2005) Article DOI: 10.1021/jm0506625 BindingDB Entry DOI: 10.7270/Q2V40SDN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||