 Found 4 hits for monomerid = 103908
Found 4 hits for monomerid = 103908 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM103908
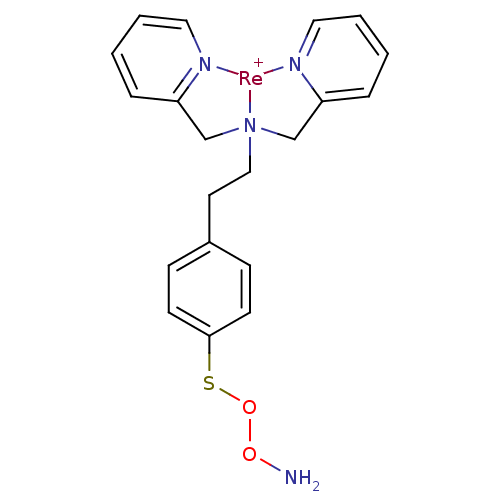
(JNK3 inhibitor 5 | US8562945, 241)Show SMILES NOOSc1ccc(CC[N]23CC4=CC=CC=[N]4[Re+]2[N]2=C(C3)C=CC=C2)cc1 |c:14,16,21,25,27,t:12| Show InChI InChI=1S/C20H22N4O2S.Re/c21-25-26-27-20-9-7-17(8-10-20)11-14-24(15-18-5-1-3-12-22-18)16-19-6-2-4-13-23-19;/h1-10,12-13H,11,14-16,21H2;/q;+1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Molecular Insight Pharmaceuticals, Inc.
US Patent
| Assay Description
Compounds were tested for their ability to inhibit carbonic anhydrase isozymes II and IX in vito. |
US Patent US8562945 (2013)
BindingDB Entry DOI: 10.7270/Q2XP73KD |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 8
(Homo sapiens (Human)) | BDBM103908

(JNK3 inhibitor 5 | US8562945, 241)Show SMILES NOOSc1ccc(CC[N]23CC4=CC=CC=[N]4[Re+]2[N]2=C(C3)C=CC=C2)cc1 |c:14,16,21,25,27,t:12| Show InChI InChI=1S/C20H22N4O2S.Re/c21-25-26-27-20-9-7-17(8-10-20)11-14-24(15-18-5-1-3-12-22-18)16-19-6-2-4-13-23-19;/h1-10,12-13H,11,14-16,21H2;/q;+1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 164 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
| |
ACS Chem Biol 8: 1747-54 (2013)
Article DOI: 10.1021/cb3006165
BindingDB Entry DOI: 10.7270/Q25719PN |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 10
(Homo sapiens (Human)) | BDBM103908

(JNK3 inhibitor 5 | US8562945, 241)Show SMILES NOOSc1ccc(CC[N]23CC4=CC=CC=[N]4[Re+]2[N]2=C(C3)C=CC=C2)cc1 |c:14,16,21,25,27,t:12| Show InChI InChI=1S/C20H22N4O2S.Re/c21-25-26-27-20-9-7-17(8-10-20)11-14-24(15-18-5-1-3-12-22-18)16-19-6-2-4-13-23-19;/h1-10,12-13H,11,14-16,21H2;/q;+1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 63.4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
| |
ACS Chem Biol 8: 1747-54 (2013)
Article DOI: 10.1021/cb3006165
BindingDB Entry DOI: 10.7270/Q25719PN |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM103908

(JNK3 inhibitor 5 | US8562945, 241)Show SMILES NOOSc1ccc(CC[N]23CC4=CC=CC=[N]4[Re+]2[N]2=C(C3)C=CC=C2)cc1 |c:14,16,21,25,27,t:12| Show InChI InChI=1S/C20H22N4O2S.Re/c21-25-26-27-20-9-7-17(8-10-20)11-14-24(15-18-5-1-3-12-22-18)16-19-6-2-4-13-23-19;/h1-10,12-13H,11,14-16,21H2;/q;+1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 445 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Molecular Insight Pharmaceuticals, Inc.
US Patent
| Assay Description
Compounds were tested for their ability to inhibit carbonic anhydrase isozymes II and IX in vito. |
US Patent US8562945 (2013)
BindingDB Entry DOI: 10.7270/Q2XP73KD |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data