

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM10478 A2A.2HCl::Bis-THA inhibitor 1::CHEMBL213377::N-[2-(1,2,3,4-tetrahydroacridin-9-ylamino)ethyl]-1,2,3,4-tetrahydroacridin-9-amine dihydrochloride
SMILES: C(CNc1c2CCCCc2nc2ccccc12)Nc1c2CCCCc2nc2ccccc12
InChI Key: InChIKey=BDUKQAPOADCBKB-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM10478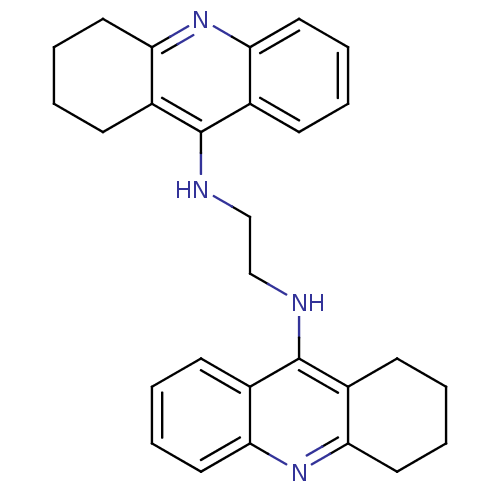 (A2A.2HCl | Bis-THA inhibitor 1 | CHEMBL213377 | N-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 711 | n/a | n/a | n/a | n/a | 7.4 | 37 |
Hong Kong University of Science and Technology | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. Enzyme activity was determined by measuring the absorbance at... | Bioorg Med Chem 7: 351-7 (1999) Article DOI: 10.1016/s0968-0896(98)00213-2 BindingDB Entry DOI: 10.7270/Q2T43R9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM10478 (A2A.2HCl | Bis-THA inhibitor 1 | CHEMBL213377 | N-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 711 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Science Curated by ChEMBL | Assay Description Inhibition of rat brain AChE | J Med Chem 49: 5491-500 (2006) Article DOI: 10.1021/jm060164b BindingDB Entry DOI: 10.7270/Q2154GPX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxylic ester hydrolase (Rattus norvegicus (rat)) | BDBM10478 (A2A.2HCl | Bis-THA inhibitor 1 | CHEMBL213377 | N-...) | Reactome pathway KEGG UniProtKB/TrEMBL GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 102 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Science Curated by ChEMBL | Assay Description Inhibition of rat serum BuChE | J Med Chem 49: 5491-500 (2006) Article DOI: 10.1021/jm060164b BindingDB Entry DOI: 10.7270/Q2154GPX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxylic ester hydrolase (Rattus norvegicus (rat)) | BDBM10478 (A2A.2HCl | Bis-THA inhibitor 1 | CHEMBL213377 | N-...) | Reactome pathway KEGG UniProtKB/TrEMBL GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 102 | n/a | n/a | n/a | n/a | 7.4 | 37 |
Hong Kong University of Science and Technology | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. Enzyme activity was determined by measuring the absorbance at... | Bioorg Med Chem 7: 351-7 (1999) Article DOI: 10.1016/s0968-0896(98)00213-2 BindingDB Entry DOI: 10.7270/Q2T43R9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||