

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM11243 AG7088::CHEMBL20210::cmdc.202100576, 24f::ethyl (2E,4S)-4-[((2R,5S)-2-(4-fluorobenzyl)-6-methyl-5-{[(5-methylisoxazol-3-yl)carbonyl]amino}-4-oxoheptanoyl)amino]-5-[(3S)-2-oxopyrrolidin-3-yl]pent-2-enoate::ethyl (2E,4S)-4-[(2R,5S)-2-[(4-fluorophenyl)methyl]-6-methyl-5-[(5-methyl-1,2-oxazol-3-yl)formamido]-4-oxoheptanamido]-5-[(3S)-2-oxopyrrolidin-3-yl]pent-2-enoate::med.21724, Compound 29
SMILES: CCOC(=O)\C=C\[C@H](C[C@@H]1CCNC1=O)NC(=O)[C@@H](CC(=O)[C@@H](NC(=O)c1cc(C)on1)C(C)C)Cc1ccc(F)cc1
InChI Key: InChIKey=CAYJBRBGZBCZKO-BHGBQCOSSA-N
PDB links: 1 PDB ID contains inhibitors having a similarity of 90% to this monomer.
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 3C-like proteinase (3CL-PRO) (Human SARS coronavirus (SARS-CoV) (Severe acute re...) | BDBM11243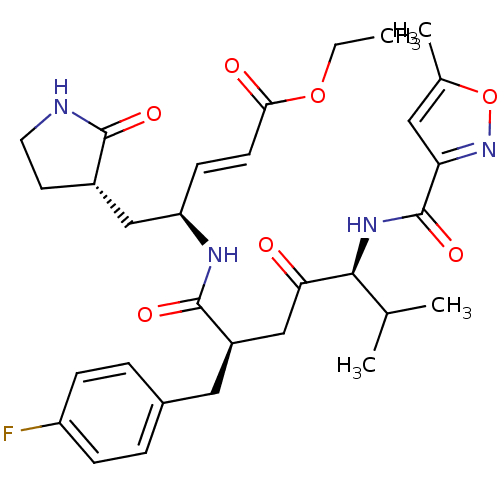 (AG7088 | CHEMBL20210 | cmdc.202100576, 24f | ethyl...) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid UniChem Patents Similars | Article PubMed | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn | Assay Description This is a review article. | Med Res Rev (2020) Article DOI: 10.1002/med.21724 BindingDB Entry DOI: 10.7270/Q2JS9ST6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Human rhinovirus A protease (Human rhinovirus B) | BDBM11243 (AG7088 | CHEMBL20210 | cmdc.202100576, 24f | ethyl...) | MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alberta Curated by ChEMBL | Assay Description Reversible inhibitory activity of the compound was determined against Protease | Bioorg Med Chem Lett 14: 3655-8 (2004) Article DOI: 10.1016/j.bmcl.2004.05.021 BindingDB Entry DOI: 10.7270/Q22R3R4V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Human rhinovirus A protease (Human rhinovirus B) | BDBM11243 (AG7088 | CHEMBL20210 | cmdc.202100576, 24f | ethyl...) | MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 5 | n/a | n/a | n/a | n/a |
Agouron Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Increased percentage of formazan production in drug treated virus infected cells to equal 50% control drug free uninfected cells on serotype 14 | J Med Chem 42: 1213-24 (1999) Article DOI: 10.1021/jm9805384 BindingDB Entry DOI: 10.7270/Q2MW2G9R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Replicase polyprotein 1ab (2019-nCoV) | BDBM11243 (AG7088 | CHEMBL20210 | cmdc.202100576, 24f | ethyl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Experimental Drug Development Centre | Assay Description Please point to the patents. | ChemMedChem (2021) | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Replicase polyprotein 1ab (HCoV-229E) | BDBM11243 (AG7088 | CHEMBL20210 | cmdc.202100576, 24f | ethyl...) | PDB MMDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn | Assay Description This is a review article. | Med Res Rev (2020) Article DOI: 10.1002/med.21724 BindingDB Entry DOI: 10.7270/Q2JS9ST6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 3C-like proteinase (3CL-PRO) (Human SARS coronavirus (SARS-CoV) (Severe acute re...) | BDBM11243 (AG7088 | CHEMBL20210 | cmdc.202100576, 24f | ethyl...) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | 7.0 | 25 |
National Taiwan University | Assay Description The effects of compound on enzyme activity were measured by using a fluorogenic peptide cleavage assay. Enhanced fluorescence caused by cleavage of t... | Bioorg Med Chem 13: 5240-52 (2005) Article DOI: 10.1016/j.bmc.2005.05.065 BindingDB Entry DOI: 10.7270/Q2VT1Q9R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 3C-like proteinase (3CL-PRO) (Human SARS coronavirus (SARS-CoV) (Severe acute re...) | BDBM11243 (AG7088 | CHEMBL20210 | cmdc.202100576, 24f | ethyl...) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 8.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn | Assay Description This is a review article. | Med Res Rev (2020) Article DOI: 10.1002/med.21724 BindingDB Entry DOI: 10.7270/Q2JS9ST6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||