

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM11530 (1S,3S,5S)-2-[(2S)-2-amino-2-(1-ethenylcyclohexyl)acetyl]-2-azabicyclo[3.1.0]hexane-3-carbonitrile; 2,2,2-trifluoroacetic acid::(S)-2-(1-Ethenylcyclohex-1-yl)glycine-L-cis-4,5-methanoprolinenitrile TFA salt::BMS-477118 analogue::Saxagliptin Analogue 8e
SMILES: N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C1(CCCCC1)C=C
InChI Key: InChIKey=VBUZSADDFRYATJ-ZOBORPQBSA-N
Data: 1 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dipeptidyl peptidase 4 (Homo sapiens (Human)) | BDBM11530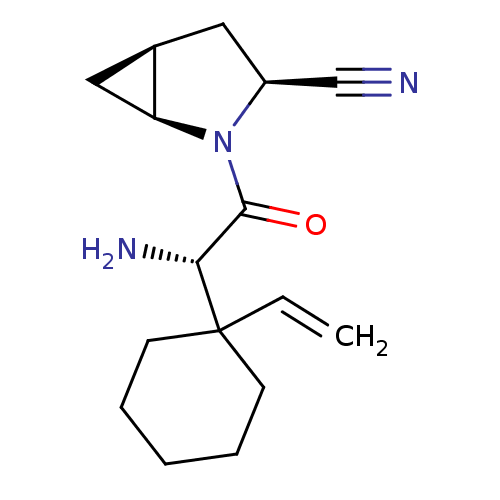 ((1S,3S,5S)-2-[(2S)-2-amino-2-(1-ethenylcyclohexyl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 1.40 | -11.9 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute | Assay Description Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... | J Med Chem 48: 5025-37 (2005) Article DOI: 10.1021/jm050261p BindingDB Entry DOI: 10.7270/Q2FN14DM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||