 Found 18 hits for monomerid = 12311
Found 18 hits for monomerid = 12311 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
HCV NS3-NS4A Serine Proteinase
(Hepatitis C virus (HCV genotype 1a, isolate H)) | BDBM12311
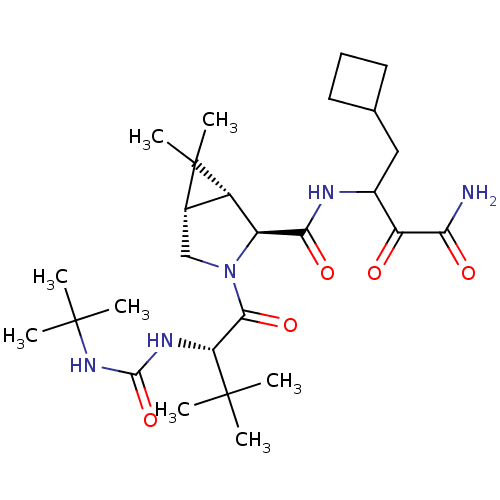
((1R,5S)-N-[3-Amino-1-(cyclobutylmethyl)-2,3-dioxop...)Show SMILES CC(C)(C)NC(=O)N[C@H](C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)NC(CC1CCC1)C(=O)C(N)=O)C2(C)C)C(C)(C)C |r| Show InChI InChI=1S/C27H45N5O5/c1-25(2,3)20(30-24(37)31-26(4,5)6)23(36)32-13-15-17(27(15,7)8)18(32)22(35)29-16(19(33)21(28)34)12-14-10-9-11-14/h14-18,20H,9-13H2,1-8H3,(H2,28,34)(H,29,35)(H2,30,31,37)/t15-,16?,17-,18-,20+/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 14 | -10.9 | n/a | n/a | n/a | n/a | n/a | 6.5 | 30 |
Schering-Plough Research Institute
| Assay Description
Proteolytic cleavage of the ester linkage between the Nva (L-norvaline) and the chromophore (PAP) was monitored for change in absorbance at 370 nm. I... |
J Med Chem 50: 2310-8 (2007)
Article DOI: 10.1021/jm060173k
BindingDB Entry DOI: 10.7270/Q27D2SDK |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
HCV NS3-NS4A Serine Proteinase
(Hepatitis C virus (HCV genotype 1a, isolate H)) | BDBM12311

((1R,5S)-N-[3-Amino-1-(cyclobutylmethyl)-2,3-dioxop...)Show SMILES CC(C)(C)NC(=O)N[C@H](C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)NC(CC1CCC1)C(=O)C(N)=O)C2(C)C)C(C)(C)C |r| Show InChI InChI=1S/C27H45N5O5/c1-25(2,3)20(30-24(37)31-26(4,5)6)23(36)32-13-15-17(27(15,7)8)18(32)22(35)29-16(19(33)21(28)34)12-14-10-9-11-14/h14-18,20H,9-13H2,1-8H3,(H2,28,34)(H,29,35)(H2,30,31,37)/t15-,16?,17-,18-,20+/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
| Assay Description
Proteolytic cleavage of the ester linkage between the Nva (L-norvaline) and the chromophore (PAP) was monitored for change in absorbance at 370 nm. I... |
J Med Chem 49: 6074-86 (2006)
Article DOI: 10.1021/jm060325b
BindingDB Entry DOI: 10.7270/Q2DF6PF6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nonstructural protein NS3-4A
(Hepatitis C virus) | BDBM12311

((1R,5S)-N-[3-Amino-1-(cyclobutylmethyl)-2,3-dioxop...)Show SMILES CC(C)(C)NC(=O)N[C@H](C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)NC(CC1CCC1)C(=O)C(N)=O)C2(C)C)C(C)(C)C |r| Show InChI InChI=1S/C27H45N5O5/c1-25(2,3)20(30-24(37)31-26(4,5)6)23(36)32-13-15-17(27(15,7)8)18(32)22(35)29-16(19(33)21(28)34)12-14-10-9-11-14/h14-18,20H,9-13H2,1-8H3,(H2,28,34)(H,29,35)(H2,30,31,37)/t15-,16?,17-,18-,20+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of HCV genotype 1a NS3/4A protease using Ac-DED(Edans)EEAbu-psi[COO]ASK(Dabcyl)-NH2 as substrate by FRET assay |
Bioorg Med Chem 22: 6595-615 (2015)
Article DOI: 10.1016/j.bmc.2014.10.010
BindingDB Entry DOI: 10.7270/Q2GX4D50 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cathepsin K
(Homo sapiens (Human)) | BDBM12311

((1R,5S)-N-[3-Amino-1-(cyclobutylmethyl)-2,3-dioxop...)Show SMILES CC(C)(C)NC(=O)N[C@H](C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)NC(CC1CCC1)C(=O)C(N)=O)C2(C)C)C(C)(C)C |r| Show InChI InChI=1S/C27H45N5O5/c1-25(2,3)20(30-24(37)31-26(4,5)6)23(36)32-13-15-17(27(15,7)8)18(32)22(35)29-16(19(33)21(28)34)12-14-10-9-11-14/h14-18,20H,9-13H2,1-8H3,(H2,28,34)(H,29,35)(H2,30,31,37)/t15-,16?,17-,18-,20+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Cathepsin K |
Antimicrob Agents Chemother 54: 305-11 (2009)
Article DOI: 10.1128/AAC.00677-09
BindingDB Entry DOI: 10.7270/Q2JW8F39 |
More data for this
Ligand-Target Pair | |
Cathepsin L2
(Homo sapiens (Human)) | BDBM12311

((1R,5S)-N-[3-Amino-1-(cyclobutylmethyl)-2,3-dioxop...)Show SMILES CC(C)(C)NC(=O)N[C@H](C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)NC(CC1CCC1)C(=O)C(N)=O)C2(C)C)C(C)(C)C |r| Show InChI InChI=1S/C27H45N5O5/c1-25(2,3)20(30-24(37)31-26(4,5)6)23(36)32-13-15-17(27(15,7)8)18(32)22(35)29-16(19(33)21(28)34)12-14-10-9-11-14/h14-18,20H,9-13H2,1-8H3,(H2,28,34)(H,29,35)(H2,30,31,37)/t15-,16?,17-,18-,20+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 75 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Cathepsin V |
Antimicrob Agents Chemother 54: 305-11 (2009)
Article DOI: 10.1128/AAC.00677-09
BindingDB Entry DOI: 10.7270/Q2JW8F39 |
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM12311

((1R,5S)-N-[3-Amino-1-(cyclobutylmethyl)-2,3-dioxop...)Show SMILES CC(C)(C)NC(=O)N[C@H](C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)NC(CC1CCC1)C(=O)C(N)=O)C2(C)C)C(C)(C)C |r| Show InChI InChI=1S/C27H45N5O5/c1-25(2,3)20(30-24(37)31-26(4,5)6)23(36)32-13-15-17(27(15,7)8)18(32)22(35)29-16(19(33)21(28)34)12-14-10-9-11-14/h14-18,20H,9-13H2,1-8H3,(H2,28,34)(H,29,35)(H2,30,31,37)/t15-,16?,17-,18-,20+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 5.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
A*STAR
| Assay Description
The assays are from references cited in this article. |
Bioorg Med Chem Lett 48: 128263 (2021)
|
More data for this
Ligand-Target Pair | |
Procathepsin L
(Homo sapiens (Human)) | BDBM12311

((1R,5S)-N-[3-Amino-1-(cyclobutylmethyl)-2,3-dioxop...)Show SMILES CC(C)(C)NC(=O)N[C@H](C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)NC(CC1CCC1)C(=O)C(N)=O)C2(C)C)C(C)(C)C |r| Show InChI InChI=1S/C27H45N5O5/c1-25(2,3)20(30-24(37)31-26(4,5)6)23(36)32-13-15-17(27(15,7)8)18(32)22(35)29-16(19(33)21(28)34)12-14-10-9-11-14/h14-18,20H,9-13H2,1-8H3,(H2,28,34)(H,29,35)(H2,30,31,37)/t15-,16?,17-,18-,20+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 760 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Cathepsin L |
Antimicrob Agents Chemother 54: 305-11 (2009)
Article DOI: 10.1128/AAC.00677-09
BindingDB Entry DOI: 10.7270/Q2JW8F39 |
More data for this
Ligand-Target Pair | |
Cathepsin F
(Homo sapiens (Human)) | BDBM12311

((1R,5S)-N-[3-Amino-1-(cyclobutylmethyl)-2,3-dioxop...)Show SMILES CC(C)(C)NC(=O)N[C@H](C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)NC(CC1CCC1)C(=O)C(N)=O)C2(C)C)C(C)(C)C |r| Show InChI InChI=1S/C27H45N5O5/c1-25(2,3)20(30-24(37)31-26(4,5)6)23(36)32-13-15-17(27(15,7)8)18(32)22(35)29-16(19(33)21(28)34)12-14-10-9-11-14/h14-18,20H,9-13H2,1-8H3,(H2,28,34)(H,29,35)(H2,30,31,37)/t15-,16?,17-,18-,20+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Cathepsin F |
Antimicrob Agents Chemother 54: 305-11 (2009)
Article DOI: 10.1128/AAC.00677-09
BindingDB Entry DOI: 10.7270/Q2JW8F39 |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM12311

((1R,5S)-N-[3-Amino-1-(cyclobutylmethyl)-2,3-dioxop...)Show SMILES CC(C)(C)NC(=O)N[C@H](C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)NC(CC1CCC1)C(=O)C(N)=O)C2(C)C)C(C)(C)C |r| Show InChI InChI=1S/C27H45N5O5/c1-25(2,3)20(30-24(37)31-26(4,5)6)23(36)32-13-15-17(27(15,7)8)18(32)22(35)29-16(19(33)21(28)34)12-14-10-9-11-14/h14-18,20H,9-13H2,1-8H3,(H2,28,34)(H,29,35)(H2,30,31,37)/t15-,16?,17-,18-,20+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >3.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Cathepsin B |
Antimicrob Agents Chemother 54: 305-11 (2009)
Article DOI: 10.1128/AAC.00677-09
BindingDB Entry DOI: 10.7270/Q2JW8F39 |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM12311

((1R,5S)-N-[3-Amino-1-(cyclobutylmethyl)-2,3-dioxop...)Show SMILES CC(C)(C)NC(=O)N[C@H](C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)NC(CC1CCC1)C(=O)C(N)=O)C2(C)C)C(C)(C)C |r| Show InChI InChI=1S/C27H45N5O5/c1-25(2,3)20(30-24(37)31-26(4,5)6)23(36)32-13-15-17(27(15,7)8)18(32)22(35)29-16(19(33)21(28)34)12-14-10-9-11-14/h14-18,20H,9-13H2,1-8H3,(H2,28,34)(H,29,35)(H2,30,31,37)/t15-,16?,17-,18-,20+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim (Canada) Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CatB after 60 mins fluorescence assay |
Antimicrob Agents Chemother 54: 4611-8 (2010)
Article DOI: 10.1128/AAC.00787-10
BindingDB Entry DOI: 10.7270/Q2NP24Q7 |
More data for this
Ligand-Target Pair | |
Leukocyte elastase
(Homo sapiens (Human)) | BDBM12311

((1R,5S)-N-[3-Amino-1-(cyclobutylmethyl)-2,3-dioxop...)Show SMILES CC(C)(C)NC(=O)N[C@H](C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)NC(CC1CCC1)C(=O)C(N)=O)C2(C)C)C(C)(C)C |r| Show InChI InChI=1S/C27H45N5O5/c1-25(2,3)20(30-24(37)31-26(4,5)6)23(36)32-13-15-17(27(15,7)8)18(32)22(35)29-16(19(33)21(28)34)12-14-10-9-11-14/h14-18,20H,9-13H2,1-8H3,(H2,28,34)(H,29,35)(H2,30,31,37)/t15-,16?,17-,18-,20+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim (Canada) Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human leukocyte elastase after 60 mins fluorescence assay |
Antimicrob Agents Chemother 54: 4611-8 (2010)
Article DOI: 10.1128/AAC.00787-10
BindingDB Entry DOI: 10.7270/Q2NP24Q7 |
More data for this
Ligand-Target Pair | |
Plasminogen
(Homo sapiens (Human)) | BDBM12311

((1R,5S)-N-[3-Amino-1-(cyclobutylmethyl)-2,3-dioxop...)Show SMILES CC(C)(C)NC(=O)N[C@H](C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)NC(CC1CCC1)C(=O)C(N)=O)C2(C)C)C(C)(C)C |r| Show InChI InChI=1S/C27H45N5O5/c1-25(2,3)20(30-24(37)31-26(4,5)6)23(36)32-13-15-17(27(15,7)8)18(32)22(35)29-16(19(33)21(28)34)12-14-10-9-11-14/h14-18,20H,9-13H2,1-8H3,(H2,28,34)(H,29,35)(H2,30,31,37)/t15-,16?,17-,18-,20+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of human plasmin |
Bioorg Med Chem Lett 23: 6325-30 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.068
BindingDB Entry DOI: 10.7270/Q2Z3213V |
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM12311

((1R,5S)-N-[3-Amino-1-(cyclobutylmethyl)-2,3-dioxop...)Show SMILES CC(C)(C)NC(=O)N[C@H](C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)NC(CC1CCC1)C(=O)C(N)=O)C2(C)C)C(C)(C)C |r| Show InChI InChI=1S/C27H45N5O5/c1-25(2,3)20(30-24(37)31-26(4,5)6)23(36)32-13-15-17(27(15,7)8)18(32)22(35)29-16(19(33)21(28)34)12-14-10-9-11-14/h14-18,20H,9-13H2,1-8H3,(H2,28,34)(H,29,35)(H2,30,31,37)/t15-,16?,17-,18-,20+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
A*STAR
| Assay Description
SARS-CoV-2 3CLpro expression and purification is based on a published procedure and our modified protocol is found in the supplementary file. A highl... |
Bioorg Med Chem Lett 48: 128263 (2021)
|
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM12311

((1R,5S)-N-[3-Amino-1-(cyclobutylmethyl)-2,3-dioxop...)Show SMILES CC(C)(C)NC(=O)N[C@H](C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)NC(CC1CCC1)C(=O)C(N)=O)C2(C)C)C(C)(C)C |r| Show InChI InChI=1S/C27H45N5O5/c1-25(2,3)20(30-24(37)31-26(4,5)6)23(36)32-13-15-17(27(15,7)8)18(32)22(35)29-16(19(33)21(28)34)12-14-10-9-11-14/h14-18,20H,9-13H2,1-8H3,(H2,28,34)(H,29,35)(H2,30,31,37)/t15-,16?,17-,18-,20+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 4.13E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
A*STAR
| Assay Description
The assays are from references cited in this article. |
Bioorg Med Chem Lett 48: 128263 (2021)
|
More data for this
Ligand-Target Pair | |
Chymase
(Homo sapiens (Human)) | BDBM12311

((1R,5S)-N-[3-Amino-1-(cyclobutylmethyl)-2,3-dioxop...)Show SMILES CC(C)(C)NC(=O)N[C@H](C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)NC(CC1CCC1)C(=O)C(N)=O)C2(C)C)C(C)(C)C |r| Show InChI InChI=1S/C27H45N5O5/c1-25(2,3)20(30-24(37)31-26(4,5)6)23(36)32-13-15-17(27(15,7)8)18(32)22(35)29-16(19(33)21(28)34)12-14-10-9-11-14/h14-18,20H,9-13H2,1-8H3,(H2,28,34)(H,29,35)(H2,30,31,37)/t15-,16?,17-,18-,20+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Chymase |
Antimicrob Agents Chemother 54: 305-11 (2009)
Article DOI: 10.1128/AAC.00677-09
BindingDB Entry DOI: 10.7270/Q2JW8F39 |
More data for this
Ligand-Target Pair | |
Leukocyte elastase
(Homo sapiens (Human)) | BDBM12311

((1R,5S)-N-[3-Amino-1-(cyclobutylmethyl)-2,3-dioxop...)Show SMILES CC(C)(C)NC(=O)N[C@H](C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)NC(CC1CCC1)C(=O)C(N)=O)C2(C)C)C(C)(C)C |r| Show InChI InChI=1S/C27H45N5O5/c1-25(2,3)20(30-24(37)31-26(4,5)6)23(36)32-13-15-17(27(15,7)8)18(32)22(35)29-16(19(33)21(28)34)12-14-10-9-11-14/h14-18,20H,9-13H2,1-8H3,(H2,28,34)(H,29,35)(H2,30,31,37)/t15-,16?,17-,18-,20+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Neutrophil elastase 2 |
Antimicrob Agents Chemother 54: 305-11 (2009)
Article DOI: 10.1128/AAC.00677-09
BindingDB Entry DOI: 10.7270/Q2JW8F39 |
More data for this
Ligand-Target Pair | |
Elastase 1
(Homo sapiens (Human)) | BDBM12311

((1R,5S)-N-[3-Amino-1-(cyclobutylmethyl)-2,3-dioxop...)Show SMILES CC(C)(C)NC(=O)N[C@H](C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)NC(CC1CCC1)C(=O)C(N)=O)C2(C)C)C(C)(C)C |r| Show InChI InChI=1S/C27H45N5O5/c1-25(2,3)20(30-24(37)31-26(4,5)6)23(36)32-13-15-17(27(15,7)8)18(32)22(35)29-16(19(33)21(28)34)12-14-10-9-11-14/h14-18,20H,9-13H2,1-8H3,(H2,28,34)(H,29,35)(H2,30,31,37)/t15-,16?,17-,18-,20+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Pancreatic elastase 1 |
Antimicrob Agents Chemother 54: 305-11 (2009)
Article DOI: 10.1128/AAC.00677-09
BindingDB Entry DOI: 10.7270/Q2JW8F39 |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM12311

((1R,5S)-N-[3-Amino-1-(cyclobutylmethyl)-2,3-dioxop...)Show SMILES CC(C)(C)NC(=O)N[C@H](C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)NC(CC1CCC1)C(=O)C(N)=O)C2(C)C)C(C)(C)C |r| Show InChI InChI=1S/C27H45N5O5/c1-25(2,3)20(30-24(37)31-26(4,5)6)23(36)32-13-15-17(27(15,7)8)18(32)22(35)29-16(19(33)21(28)34)12-14-10-9-11-14/h14-18,20H,9-13H2,1-8H3,(H2,28,34)(H,29,35)(H2,30,31,37)/t15-,16?,17-,18-,20+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 120 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Cathepsin S |
Antimicrob Agents Chemother 54: 305-11 (2009)
Article DOI: 10.1128/AAC.00677-09
BindingDB Entry DOI: 10.7270/Q2JW8F39 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data