

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM13126 (2R)-2-[benzyl(4-methoxybenzene)sulfonamido]-N-hydroxy-3-methylbutanamide::CGS 27023A Analog 58::CHEMBL85030
SMILES: COc1ccc(cc1)S(=O)(=O)N(Cc1ccccc1)[C@H](C(C)C)C(=O)NO
InChI Key: InChIKey=ZGSTVUAZFACDLH-GOSISDBHSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM13126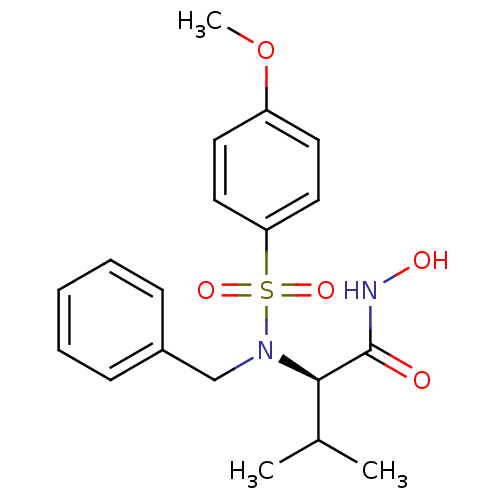 ((2R)-2-[benzyl(4-methoxybenzene)sulfonamido]-N-hyd...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University Hospital M£nster Curated by ChEMBL | Assay Description Inhibition of MMP-2 (unknown origin) | J Med Chem 56: 6858-70 (2013) Article DOI: 10.1021/jm4006753 BindingDB Entry DOI: 10.7270/Q2TF018J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM13126 ((2R)-2-[benzyl(4-methoxybenzene)sulfonamido]-N-hyd...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Westf£lische Wilhelms-Universit£t M£nster Curated by ChEMBL | Assay Description Inhibition of human active MMP9 using (7-methoxycoumarin-4-yl) acetyl pro-Leu-Gly-Leu-[3-(2,4-dinitrophenyl)-L-2,3-diamino-propionyl]-Ala-Arg-NH2 flu... | Bioorg Med Chem 23: 3809-18 (2015) Article DOI: 10.1016/j.bmc.2015.03.078 BindingDB Entry DOI: 10.7270/Q2NS0WNQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM13126 ((2R)-2-[benzyl(4-methoxybenzene)sulfonamido]-N-hyd...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University Hospital M£nster Curated by ChEMBL | Assay Description Inhibition of MMP2 using (7-methoxycoumarin-4-yl)acetyl-Pro-Leu-Gly-Leu-(3-(2,4-dinitrophenyl)-L-2,3-diaminopropionyl)Ala-Arg-NH2 as substrate incuba... | J Med Chem 55: 4714-27 (2012) Article DOI: 10.1021/jm300199g BindingDB Entry DOI: 10.7270/Q2GQ6ZTT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM13126 ((2R)-2-[benzyl(4-methoxybenzene)sulfonamido]-N-hyd...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 23 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University Hospital M£nster Curated by ChEMBL | Assay Description Inhibition of MMP8 using (7-methoxycoumarin-4-yl)acetyl-Pro-Leu-Gly-Leu-(3-(2,4-dinitrophenyl)-L-2,3-diaminopropionyl)Ala-Arg-NH2 as substrate incuba... | J Med Chem 55: 4714-27 (2012) Article DOI: 10.1021/jm300199g BindingDB Entry DOI: 10.7270/Q2GQ6ZTT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM13126 ((2R)-2-[benzyl(4-methoxybenzene)sulfonamido]-N-hyd...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 23 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University Hospital M£nster Curated by ChEMBL | Assay Description Inhibition of MMP-8 (unknown origin) | J Med Chem 56: 6858-70 (2013) Article DOI: 10.1021/jm4006753 BindingDB Entry DOI: 10.7270/Q2TF018J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM13126 ((2R)-2-[benzyl(4-methoxybenzene)sulfonamido]-N-hyd...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University Hospital M£nster Curated by ChEMBL | Assay Description Inhibition of MMP9 using (7-methoxycoumarin-4-yl)acetyl-Pro-Leu-Gly-Leu-(3-(2,4-dinitrophenyl)-L-2,3-diaminopropionyl)Ala-Arg-NH2 as substrate incuba... | J Med Chem 55: 4714-27 (2012) Article DOI: 10.1021/jm300199g BindingDB Entry DOI: 10.7270/Q2GQ6ZTT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM13126 ((2R)-2-[benzyl(4-methoxybenzene)sulfonamido]-N-hyd...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Westf£lische Wilhelms-Universit£t M£nster Curated by ChEMBL | Assay Description Inhibition of human active MMP9 using (7-methoxycoumarin-4-yl) acetyl pro-Leu-Gly-Leu-[3-(2,4-dinitrophenyl)-L-2,3-diamino-propionyl]-Ala-Arg-NH2 flu... | Bioorg Med Chem 23: 3809-18 (2015) Article DOI: 10.1016/j.bmc.2015.03.078 BindingDB Entry DOI: 10.7270/Q2NS0WNQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM13126 ((2R)-2-[benzyl(4-methoxybenzene)sulfonamido]-N-hyd...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University Hospital M£nster Curated by ChEMBL | Assay Description Inhibition of MMP-9 (unknown origin) | J Med Chem 56: 6858-70 (2013) Article DOI: 10.1021/jm4006753 BindingDB Entry DOI: 10.7270/Q2TF018J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Stromelysin-1 (Homo sapiens (Human)) | BDBM13126 ((2R)-2-[benzyl(4-methoxybenzene)sulfonamido]-N-hyd...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 34 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals | Assay Description Stromelysin inhibitory activity is based on the hydrolysis of substance P by recombinant human stromelysin to generate a fragment, substance P 7-11, ... | J Med Chem 40: 2525-32 (1997) Article DOI: 10.1021/jm960871c BindingDB Entry DOI: 10.7270/Q2MW2FC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase 12 (Mus musculus) | BDBM13126 ((2R)-2-[benzyl(4-methoxybenzene)sulfonamido]-N-hyd...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 4.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals Curated by ChEMBL | Assay Description Inhibitory activity against mouse macrophage metalloelastase (MME) using [3H]elastin as a substrate | Bioorg Med Chem Lett 8: 897-902 (1999) BindingDB Entry DOI: 10.7270/Q2NP24ZC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||