

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM13592 2-{2-[(4-carbamimidoylphenyl)carbamoyl]-6-methoxypyridin-3-yl}-5-{[(2S)-1-hydroxy-3,3-dimethylbutan-2-yl]carbamoyl}benzoic acid::Ono Compound 8
SMILES: COc1ccc(-c2ccc(cc2C(O)=O)C(=O)N[C@H](CO)C(C)(C)C)c(n1)C(=O)Nc1ccc(cc1)C(N)=N
InChI Key: InChIKey=KPACBFJTZSMBKD-OAQYLSRUSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Coagulation factor VII (Homo sapiens (Human)) | BDBM13592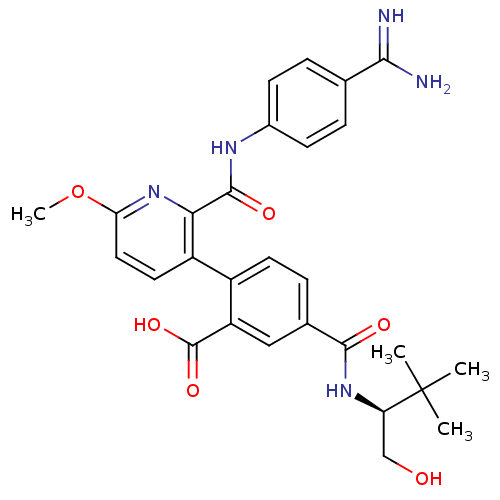 (2-{2-[(4-carbamimidoylphenyl)carbamoyl]-6-methoxyp...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 10 | -10.8 | n/a | n/a | n/a | n/a | n/a | 7.8 | 22 |
F. Hoffmann-La Roche Ltd. | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem 14: 5357-69 (2006) Article DOI: 10.1016/j.bmc.2006.03.042 BindingDB Entry DOI: 10.7270/Q2NP22PZ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM13592 (2-{2-[(4-carbamimidoylphenyl)carbamoyl]-6-methoxyp...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd. | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem 14: 5357-69 (2006) Article DOI: 10.1016/j.bmc.2006.03.042 BindingDB Entry DOI: 10.7270/Q2NP22PZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Trypsin (Bos taurus (bovine)) | BDBM13592 (2-{2-[(4-carbamimidoylphenyl)carbamoyl]-6-methoxyp...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB Article PubMed | 240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd. | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem 14: 5357-69 (2006) Article DOI: 10.1016/j.bmc.2006.03.042 BindingDB Entry DOI: 10.7270/Q2NP22PZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM13592 (2-{2-[(4-carbamimidoylphenyl)carbamoyl]-6-methoxyp...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 4.30E+3 | -7.24 | n/a | n/a | n/a | n/a | n/a | 7.8 | 22 |
F. Hoffmann-La Roche Ltd. | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem 14: 5357-69 (2006) Article DOI: 10.1016/j.bmc.2006.03.042 BindingDB Entry DOI: 10.7270/Q2NP22PZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM13592 (2-{2-[(4-carbamimidoylphenyl)carbamoyl]-6-methoxyp...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 35 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development Curated by ChEMBL | Assay Description Inhibition of human F10a using S-2222 as substrate after 3 mins | Bioorg Med Chem Lett 23: 5239-43 (2013) Article DOI: 10.1016/j.bmcl.2013.06.028 BindingDB Entry DOI: 10.7270/Q2CC123C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor XI (Homo sapiens (Human)) | BDBM13592 (2-{2-[(4-carbamimidoylphenyl)carbamoyl]-6-methoxyp...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 3.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development Curated by ChEMBL | Assay Description Inhibition of human F11a using S-2366 as substrate after 3 mins | Bioorg Med Chem Lett 23: 5239-43 (2013) Article DOI: 10.1016/j.bmcl.2013.06.028 BindingDB Entry DOI: 10.7270/Q2CC123C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tissue-type plasminogen activator (Homo sapiens (Human)) | BDBM13592 (2-{2-[(4-carbamimidoylphenyl)carbamoyl]-6-methoxyp...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | >3.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development Curated by ChEMBL | Assay Description Inhibition of human tissue plasminogen activator using spectrozyme tissue plasminogen activator as substrate after 3 mins | Bioorg Med Chem Lett 23: 5239-43 (2013) Article DOI: 10.1016/j.bmcl.2013.06.028 BindingDB Entry DOI: 10.7270/Q2CC123C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM13592 (2-{2-[(4-carbamimidoylphenyl)carbamoyl]-6-methoxyp...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development Curated by ChEMBL | Assay Description Inhibition of human F7a using D-Ile-Pro-Arg-AFC as substrate after 3 mins | Bioorg Med Chem Lett 23: 5239-43 (2013) Article DOI: 10.1016/j.bmcl.2013.06.028 BindingDB Entry DOI: 10.7270/Q2CC123C | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||