

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM13775 (1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-hydroxyacetyl)-2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-6-en-5-one::11beta,17alpha,21-Trihydroxy-4-pregnene-3,20-dione::3H-cortisol::HYDROCORTISONE::US10188667, Example 00023::[3H]cortisol::cortisol
SMILES: [H][C@@]12CC[C@](O)(C(=O)CO)[C@@]1(C)C[C@H](O)[C@@]1([H])[C@@]2([H])CCC2=CC(=O)CC[C@]12C
InChI Key: InChIKey=JYGXADMDTFJGBT-VWUMJDOOSA-N
PDB links: 11 PDB IDs match this monomer. 1 PDB ID contains this monomer as substructures. 1 PDB ID contains inhibitors having a similarity of 90% to this monomer.
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Corticosteroid-binding globulin (Homo sapiens) | BDBM13775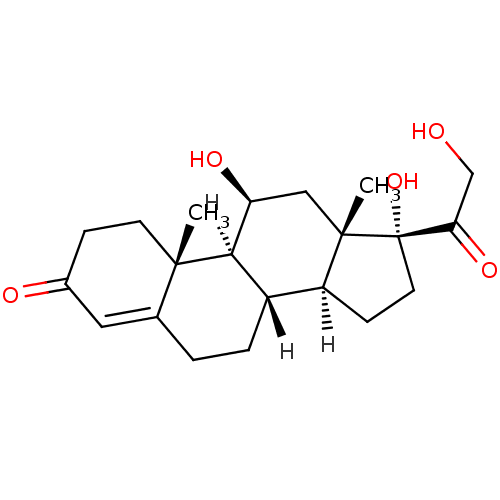 ((1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase DrugBank KEGG PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Zoki Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Binding affinity to human CBG receptor (corticosteroid-binding globulins) | J Med Chem 47: 2732-42 (2004) Article DOI: 10.1021/jm030364c BindingDB Entry DOI: 10.7270/Q2WM1H5Q | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Glucocorticoid receptor (Homo sapiens (Human)) | BDBM13775 ((1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-...) | PDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank KEGG PC cid PC sid PDB UniChem Similars | DrugBank PDB Article PubMed | 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Trinity College Curated by ChEMBL | Assay Description Displacement of [3H]DEX from human glucocorticoid receptor | J Med Chem 53: 3065-74 (2010) Article DOI: 10.1021/jm901452y BindingDB Entry DOI: 10.7270/Q2VQ33NH | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Organic anion-transporting polypeptide 1D1 (Oatp1d1) (Danio rerio (Zebrafish)) | BDBM13775 ((1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-...) | GoogleScholar AffyNet | Purchase DrugBank KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | 1.20E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rudjer Boskovic Institute | Assay Description In the inhibition experiments, the cells were preincubated for 20 s with test compounds, followed by a 5-min incubation with [3H]E3S (5 nM) or 30-min... | J Biol Chem 288: 33894-911 (2013) Article DOI: 10.1074/jbc.M113.518506 BindingDB Entry DOI: 10.7270/Q29Z93RK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-glycoprotein 1 (Homo sapiens (Human)) | BDBM13775 ((1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | Purchase DrugBank KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-LaRoche Ltd. Curated by ChEMBL | Assay Description Inhibition of P-glycoprotein, human L-MDR1 expressed in LLC-PK1 epithelial cells using calcein-AM polarisation assay | J Med Chem 46: 1716-25 (2003) Article DOI: 10.1021/jm021012t BindingDB Entry DOI: 10.7270/Q2ZS2X8R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-glycoprotein 1 and 3 (MDR1a/MDR1b) (Mus musculus) | BDBM13775 ((1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-...) | Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL GoogleScholar AffyNet | Purchase DrugBank KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-LaRoche Ltd. Curated by ChEMBL | Assay Description Inhibition of P-glycoprotein, mouse L-mdr1b expressed in LLC-PK1 epithelial cells using calcein-AM polarisation assay | J Med Chem 46: 1716-25 (2003) Article DOI: 10.1021/jm021012t BindingDB Entry DOI: 10.7270/Q2ZS2X8R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 3A4 (Homo sapiens (Human)) | BDBM13775 ((1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank KEGG PC cid PC sid PDB UniChem Similars | DrugBank Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-LaRoche Ltd. Curated by ChEMBL | Assay Description Inhibition of human cytochrome P450 3A4 | J Med Chem 46: 1716-25 (2003) Article DOI: 10.1021/jm021012t BindingDB Entry DOI: 10.7270/Q2ZS2X8R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Testis-specific androgen-binding protein (Homo sapiens (Human)) | BDBM13775 ((1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-...) | PDB MMDB NCI pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank KEGG PC cid PC sid PDB UniChem Similars | DrugBank Article PubMed | n/a | n/a | n/a | 631 | n/a | n/a | n/a | n/a | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Displacement of [3H]5alpha dihydrotestosterone from human sex hormone binding globulin | J Med Chem 51: 2047-56 (2008) Article DOI: 10.1021/jm7011485 BindingDB Entry DOI: 10.7270/Q2RX9DC2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Rattus norvegicus (Rat)) | BDBM13775 ((1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase DrugBank KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 1.82E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Basel Curated by ChEMBL | Assay Description Inhibitory concentration against recombinant rat androgen receptor expressed in Escherichia coli using [3H]methyltrienolone (R 1881) | J Med Chem 48: 5666-74 (2005) Article DOI: 10.1021/jm050403f BindingDB Entry DOI: 10.7270/Q2TM7CBZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-glycoprotein 1 (Homo sapiens (Human)) | BDBM13775 ((1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | Purchase DrugBank KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-LaRoche Ltd. Curated by ChEMBL | Assay Description TP_TRANSPORTER: inhibition of Calcein-AM efflux in MDR1-expressing LLC-PK1 cells | J Med Chem 46: 1716-25 (2003) Article DOI: 10.1021/jm021012t BindingDB Entry DOI: 10.7270/Q2ZS2X8R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Rho GTPase-activating protein 35 (Homo sapiens (Human)) | BDBM13775 ((1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase DrugBank KEGG PC cid PC sid PDB UniChem Similars | US Patent | n/a | n/a | n/a | n/a | 41.6 | n/a | n/a | n/a | n/a |
ALLERGAN, INC. US Patent | Assay Description Glucocorticoid receptor (GR) activation potency was assessed using a HeLa cell line containing the MMTV-bla reporter (MMTV-bla HeLa CELLSENSOR�, Invi... | US Patent US10188667 (2019) BindingDB Entry DOI: 10.7270/Q24Q7X38 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mineralocorticoid receptor (Homo sapiens (Human)) | BDBM13775 ((1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-...) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank KEGG PC cid PC sid PDB UniChem Similars | US Patent | n/a | n/a | n/a | n/a | 2.90 | n/a | n/a | n/a | n/a |
ALLERGAN, INC. US Patent | Assay Description Mineralocorticoid receptor (MR) activation potency was assessed using a HEK 293T cell line containing the UAS-bla reporter (UAS-bla HEK 293T CELLSENS... | US Patent US10188667 (2019) BindingDB Entry DOI: 10.7270/Q24Q7X38 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mineralocorticoid receptor (Homo sapiens (Human)) | BDBM13775 ((1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-...) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a |
Instituto de Qu£mica M£dica (IQM-CSIC) Curated by ChEMBL | Assay Description Binding affinity to MR (unknown origin) | J Med Chem 60: 2629-2650 (2017) Article DOI: 10.1021/acs.jmedchem.6b01065 BindingDB Entry DOI: 10.7270/Q2HD7XXB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Bile salt export pump (Homo sapiens (Human)) | BDBM13775 ((1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase DrugBank KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 2.52E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Curated by ChEMBL | Assay Description Inhibition of human BSEP expressed in baculovirus transfected fall armyworm Sf21 cell membranes vesicles assessed as reduction in ATP-dependent [3H]-... | Drug Metab Dispos 40: 2332-41 (2012) Article DOI: 10.1124/dmd.112.047068 BindingDB Entry DOI: 10.7270/Q2ZP488M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-glycoprotein (P-gp) (Mus musculus (Mouse)) | BDBM13775 ((1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase DrugBank KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-LaRoche Ltd. Curated by ChEMBL | Assay Description Inhibition of P-glycoprotein, mouse L-mdr1a expressed in LLC-PK1 epithelial cells using calcein-AM polarisation assay | J Med Chem 46: 1716-25 (2003) Article DOI: 10.1021/jm021012t BindingDB Entry DOI: 10.7270/Q2ZS2X8R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-glycoprotein 1 (Homo sapiens (Human)) | BDBM13775 ((1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | Purchase DrugBank KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-LaRoche Ltd. Curated by ChEMBL | Assay Description Inhibition of P-glycoprotein using calcein-AM assay transfected in porcine PBCEC | J Med Chem 46: 1716-25 (2003) Article DOI: 10.1021/jm021012t BindingDB Entry DOI: 10.7270/Q2ZS2X8R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2J2 (Homo sapiens (Human)) | BDBM13775 ((1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase DrugBank KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tongji University Curated by ChEMBL | Assay Description Inhibition of human recombinant CYP2J2 assessed as reduction in astemizole O-demethylation by LC-MS/MS method | Drug Metab Dispos 41: 60-71 (2012) Article DOI: 10.1124/dmd.112.048264 BindingDB Entry DOI: 10.7270/Q2K0761G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glucocorticoid (MOUSE) | BDBM13775 ((1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase DrugBank KEGG PC cid PC sid PDB UniChem Similars | PDB PubMed | n/a | n/a | n/a | 43 | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Displacement of 1 x 10'-8 M of [1,2,3-3H]-triamcinolone acetonide from glucocorticoid receptor in soluble fraction of mouse L929 cells after 20 hrs | J Med Chem 20: 1134-9 (1978) BindingDB Entry DOI: 10.7270/Q2RX9DM6 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||