

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM13823 2,2,2-trifluoroacetate; 4-[({1-[(3-carbamimidoylphenyl)methyl]-3-methyl-1H-indol-2-yl}formamido)methyl]-1-methylpyridin-1-ium::3-amidinobenzylindole carboxamide 7
SMILES: Cc1c(C(=O)NCc2cc[n+](C)cc2)n(Cc2cccc(c2)C(N)=N)c2ccccc12
InChI Key: InChIKey=JOBDMJBFUZYBIW-UHFFFAOYSA-O
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Coagulation factor X (Homo sapiens (Human)) | BDBM13823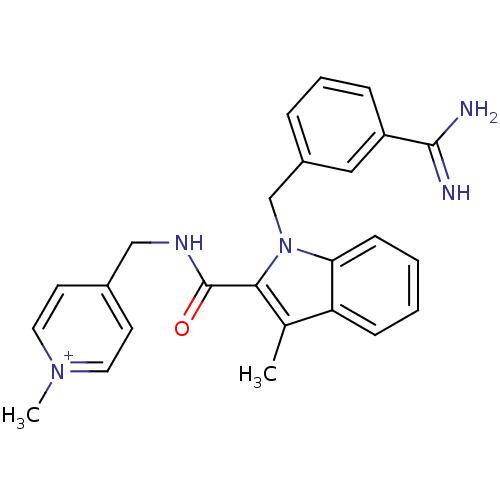 (2,2,2-trifluoroacetate; 4-[({1-[(3-carbamimidoylph...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 170 | -9.23 | n/a | n/a | n/a | n/a | n/a | 7.8 | 25 |
Aventis Pharma Deutschland GmbH | Assay Description The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrates S-2765. The hydrolysis rates of chromogenic s... | J Med Chem 45: 2749-69 (2002) Article DOI: 10.1021/jm0111346 BindingDB Entry DOI: 10.7270/Q27H1GTW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||