

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM14743 (R)-4-[3-(2-Chloro-phenoxy)-pyrrolidin-1-yl]-6,7-dimethoxy-quinazoline::4-[(3R)-3-(2-chlorophenoxy)pyrrolidin-1-yl]-6,7-dimethoxyquinazoline::6,7-Dimethoxy-4-pyrrolidylquinazoline 6
SMILES: COc1cc2ncnc(N3CC[C@H](C3)Oc3ccccc3Cl)c2cc1OC
InChI Key: InChIKey=IDUQTNNDVLFGOC-CYBMUJFWSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Phosphodiesterase 3 (PDE3) (Homo sapiens (Human)) | BDBM14743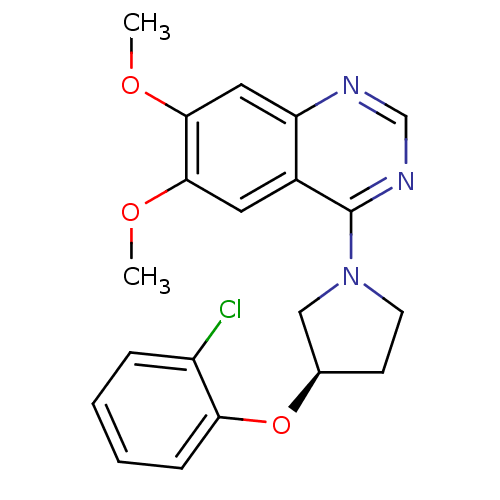 ((R)-4-[3-(2-Chloro-phenoxy)-pyrrolidin-1-yl]-6,7-d...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | <100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer | Assay Description PDE activity was monitored by measuring the hydrolysis of [3H]-cAMP to [3H]-AMP using a scintillation proximity assay (SPA). [3H]-AMP was captured by... | J Med Chem 50: 182-5 (2007) Article DOI: 10.1021/jm060653b BindingDB Entry DOI: 10.7270/Q2930RDX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cGMP-inhibited 3',5'-cyclic phosphodiesterase B (Homo sapiens (Human)) | BDBM14743 ((R)-4-[3-(2-Chloro-phenoxy)-pyrrolidin-1-yl]-6,7-d...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | <100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer | Assay Description PDE activity was monitored by measuring the hydrolysis of [3H]-cAMP to [3H]-AMP using a scintillation proximity assay (SPA). [3H]-AMP was captured by... | J Med Chem 50: 182-5 (2007) Article DOI: 10.1021/jm060653b BindingDB Entry DOI: 10.7270/Q2930RDX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A (Homo sapiens (Human)) | BDBM14743 ((R)-4-[3-(2-Chloro-phenoxy)-pyrrolidin-1-yl]-6,7-d...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 198 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
St. John's University Curated by ChEMBL | Assay Description Inhibition of PDE10A | Bioorg Med Chem 16: 3675-86 (2008) Article DOI: 10.1016/j.bmc.2008.02.013 BindingDB Entry DOI: 10.7270/Q2V988ZC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Phosphodiesterase Type 10 (PDE10A) (Rattus norvegicus (rat)) | BDBM14743 ((R)-4-[3-(2-Chloro-phenoxy)-pyrrolidin-1-yl]-6,7-d...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 198 | -9.05 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
Pfizer | Assay Description PDE activity was monitored by measuring the hydrolysis of [3H]-cAMP to [3H]-AMP using a scintillation proximity assay (SPA). [3H]-AMP was captured by... | J Med Chem 50: 182-5 (2007) Article DOI: 10.1021/jm060653b BindingDB Entry DOI: 10.7270/Q2930RDX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||