

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM1518 2-ethyl-9-methyl-2,4,9,15-tetraazatricyclo[9.4.0.0^{3,8}]pentadeca-1(11),3,5,7,12,14-hexaen-10-one::CHEMBL37343::Dipyridodiazepinone deriv. 3
SMILES: CCN1c2ncccc2N(C)C(=O)c2cccnc12
InChI Key: InChIKey=SVFKQHLOBRUZGV-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HIV-1 Reverse Transcriptase (Human immunodeficiency virus type 1) | BDBM1518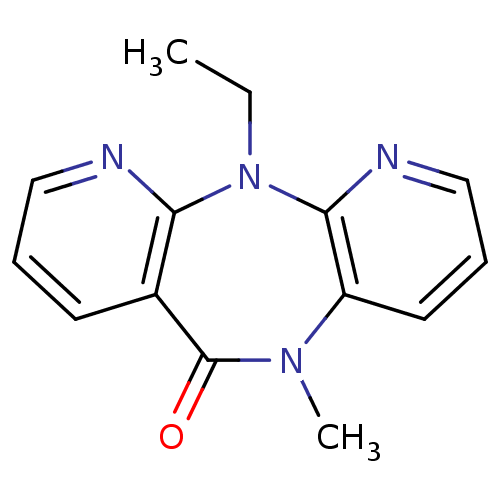 (2-ethyl-9-methyl-2,4,9,15-tetraazatricyclo[9.4.0.0...) | PDB MMDB B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 90 | n/a | n/a | n/a | n/a | 7.8 | 22 |
Boehringer Ingelheim Pharmaceuticals Inc. | Assay Description The IC50 of reverse transcriptase is the concentration that inhibits 50% of recombinant HIV-1 RT RNA-directed DNA polymerase activity in vitro. | J Med Chem 41: 2960-71 (1998) Article DOI: 10.1021/jm9707028 BindingDB Entry DOI: 10.7270/Q28W3BH2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| HIV-1 Reverse Transcriptase (Human immunodeficiency virus type 1) | BDBM1518 (2-ethyl-9-methyl-2,4,9,15-tetraazatricyclo[9.4.0.0...) | PDB MMDB B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 130 | n/a | n/a | n/a | n/a | 7.8 | 25 |
Boehringer Ingelheim Pharmaceuticals Inc. | Assay Description The IC50 of reverse transcriptase is the concentration that inhibits 50% of recombinant HIV-1 RT RNA-directed DNA polymerase activity in vitro. | J Med Chem 38: 4830-8 (1995) Article DOI: 10.1021/jm00024a010 BindingDB Entry DOI: 10.7270/Q23B5XBJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| HIV-1 Reverse Transcriptase Mutant (Y181C) (Human immunodeficiency virus type 1) | BDBM1518 (2-ethyl-9-methyl-2,4,9,15-tetraazatricyclo[9.4.0.0...) | PDB MMDB B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.20E+3 | n/a | n/a | n/a | n/a | 7.8 | 25 |
Boehringer Ingelheim Pharmaceuticals Inc. | Assay Description The IC50 of reverse transcriptase is the concentration that inhibits 50% of recombinant HIV-1 RT RNA-directed DNA polymerase activity in vitro. | J Med Chem 38: 4830-8 (1995) Article DOI: 10.1021/jm00024a010 BindingDB Entry DOI: 10.7270/Q23B5XBJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase (Human immunodeficiency virus 1) | BDBM1518 (2-ethyl-9-methyl-2,4,9,15-tetraazatricyclo[9.4.0.0...) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 125 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn Curated by ChEMBL | Assay Description Inhibition of HIV1 reverse transcriptase derived from HIV patient plasma by LC/MS/MS analysis | J Med Chem 62: 4851-4883 (2019) Article DOI: 10.1021/acs.jmedchem.8b00843 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Human immunodeficiency virus type 1 reverse transcriptase (Human immunodeficiency virus 1) | BDBM1518 (2-ethyl-9-methyl-2,4,9,15-tetraazatricyclo[9.4.0.0...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article | n/a | n/a | 125 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 Reverse transcriptase(RT) was determined | Bioorg Med Chem Lett 5: 163-166 (1995) Article DOI: 10.1016/0960-894X(94)00478-X BindingDB Entry DOI: 10.7270/Q22R3RM4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Human immunodeficiency virus type 1 reverse transcriptase (Human immunodeficiency virus 1) | BDBM1518 (2-ethyl-9-methyl-2,4,9,15-tetraazatricyclo[9.4.0.0...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 125 | n/a | n/a | n/a | n/a | n/a | n/a |
Yale University Curated by ChEMBL | Assay Description Inhibition of HIV-1 reverse transcriptase at 37 degree Centigrade | J Med Chem 44: 145-54 (2001) BindingDB Entry DOI: 10.7270/Q2VX0FRX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Human immunodeficiency virus type 1 reverse transcriptase (Human immunodeficiency virus 1) | BDBM1518 (2-ethyl-9-methyl-2,4,9,15-tetraazatricyclo[9.4.0.0...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim Pharmaceuticals Curated by ChEMBL | Assay Description In vitro for inhibition of HIV-1 reverse transcriptase. | J Med Chem 38: 1406-10 (1995) BindingDB Entry DOI: 10.7270/Q2MG7NJQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| HIV-1 Reverse Transcriptase (Human immunodeficiency virus type 1) | BDBM1518 (2-ethyl-9-methyl-2,4,9,15-tetraazatricyclo[9.4.0.0...) | PDB MMDB B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 125 | n/a | n/a | n/a | n/a | 7.8 | 22 |
Boehringer Ingelheim Pharmaceuticals Inc. | Assay Description The IC50 of reverse transcriptase is the concentration that inhibits 50% of recombinant HIV-1 RT RNA-directed DNA polymerase activity in vitro. | J Med Chem 34: 2231-41 (1991) Article DOI: 10.1021/jm00111a045 BindingDB Entry DOI: 10.7270/Q2TT4P4X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||