

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM164 (4R,5R,6R)-4-benzyl-5-hydroxy-1,3-bis(1H-indazol-5-ylmethyl)-6-(2-phenylethyl)-1,3-diazinan-2-one::CHEMBL288254::Cyclic Urea::[4R-(4,5,6)]-Tetrahydro-5-hydroxy-1,3-bis(1H-indazol-5-ylmethyl)-4-(2-phenylethyl)-6-(phenylmethyl)-2(1H)-pyrimidinone
SMILES: O[C@@H]1[C@@H](CCc2ccccc2)N(Cc2ccc3[nH]ncc3c2)C(=O)N(Cc2ccc3[nH]ncc3c2)[C@@H]1Cc1ccccc1
InChI Key: InChIKey=XYUHJAIPECDEBR-CKOYEXALSA-N
Data: 5 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Human immunodeficiency virus type 1 protease (Human immunodeficiency virus type 1) | BDBM164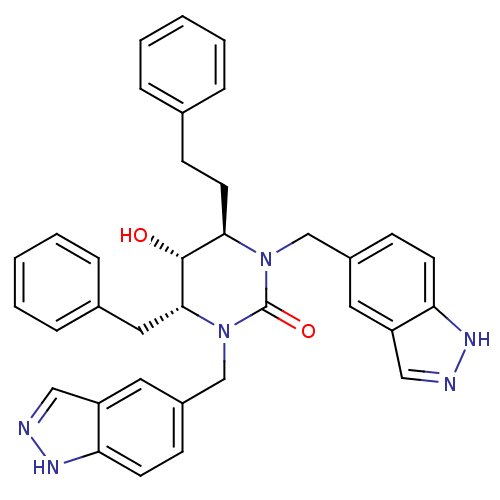 ((4R,5R,6R)-4-benzyl-5-hydroxy-1,3-bis(1H-indazol-5...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Missouri-St. Louis Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 protease | J Med Chem 45: 973-83 (2002) Article DOI: 10.1021/jm010417v BindingDB Entry DOI: 10.7270/Q2JH3PX8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Human immunodeficiency virus type 1 protease (Human immunodeficiency virus type 1) | BDBM164 ((4R,5R,6R)-4-benzyl-5-hydroxy-1,3-bis(1H-indazol-5...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company | Assay Description Inhibition constant of HIV protease inhibitors | J Med Chem 42: 135-52 (1999) Article DOI: 10.1021/jm9803626 BindingDB Entry DOI: 10.7270/Q28050S9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Human immunodeficiency virus type 1 protease (Human immunodeficiency virus type 1) | BDBM164 ((4R,5R,6R)-4-benzyl-5-hydroxy-1,3-bis(1H-indazol-5...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Florida Curated by ChEMBL | Assay Description Inhibitory activity of compound against HIV-1 aspartyl protease. | Bioorg Med Chem Lett 12: 3453-7 (2002) BindingDB Entry DOI: 10.7270/Q2B27WG3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| HIV-1 Protease (Human immunodeficiency virus type 1) | BDBM164 ((4R,5R,6R)-4-benzyl-5-hydroxy-1,3-bis(1H-indazol-5...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0230 | -15.1 | n/a | n/a | n/a | n/a | n/a | 5.5 | 37 |
DuPont Merck Pharmaceutical Company | J Med Chem 39: 3514-25 (1996) Article DOI: 10.1021/jm9602571 BindingDB Entry DOI: 10.7270/Q2ZW1J3M | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Human immunodeficiency virus type 1 protease (Human immunodeficiency virus type 1) | BDBM164 ((4R,5R,6R)-4-benzyl-5-hydroxy-1,3-bis(1H-indazol-5...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dupont Merck Pharmaceutical Company Curated by ChEMBL | Assay Description Inhibitory activity against HIV protease | J Med Chem 41: 2411-23 (1998) Article DOI: 10.1021/jm980103g BindingDB Entry DOI: 10.7270/Q2H41QJW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||