 Found 16 hits for monomerid = 17277
Found 16 hits for monomerid = 17277 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Coagulation factor X
(Homo sapiens (Human)) | BDBM17277
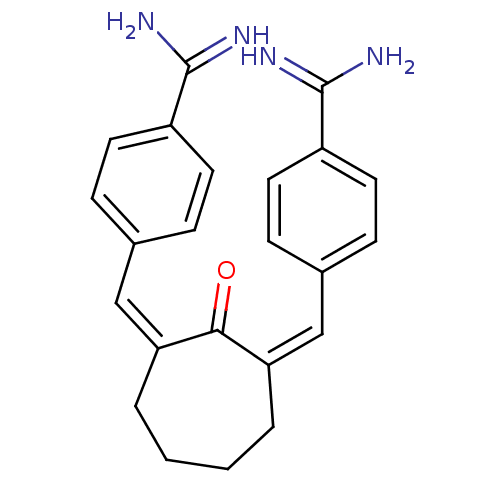
((Z,Z)BABCH | 4-{[(1Z,3Z)-3-[(4-carbamimidoylphenyl...)Show SMILES NC(=N)c1ccc(\C=C2\CCCC\C(=C\c3ccc(cc3)C(N)=N)C2=O)cc1 Show InChI InChI=1S/C23H24N4O/c24-22(25)17-9-5-15(6-10-17)13-19-3-1-2-4-20(21(19)28)14-16-7-11-18(12-8-16)23(26)27/h5-14H,1-4H2,(H3,24,25)(H3,26,27)/b19-13-,20-14- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 0.660 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Compound was tested for inhibitory activity against human Coagulation factor X |
Bioorg Med Chem Lett 8: 2235-40 (1999)
BindingDB Entry DOI: 10.7270/Q2G44PGD |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM17277

((Z,Z)BABCH | 4-{[(1Z,3Z)-3-[(4-carbamimidoylphenyl...)Show SMILES NC(=N)c1ccc(\C=C2\CCCC\C(=C\c3ccc(cc3)C(N)=N)C2=O)cc1 Show InChI InChI=1S/C23H24N4O/c24-22(25)17-9-5-15(6-10-17)13-19-3-1-2-4-20(21(19)28)14-16-7-11-18(12-8-16)23(26)27/h5-14H,1-4H2,(H3,24,25)(H3,26,27)/b19-13-,20-14- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.660 | -12.4 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
Berlex
| Assay Description
The enzyme activities were determined kinetically as the initial rate of cleavage of a peptide p-nitroanilide. Km for enzyme and substrate was determ... |
Acta Crystallogr D Biol Crystallogr 55: 1395-404 (1999)
Article DOI: 10.1107/s0907444999007350
BindingDB Entry DOI: 10.7270/Q2H1308B |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM17277

((Z,Z)BABCH | 4-{[(1Z,3Z)-3-[(4-carbamimidoylphenyl...)Show SMILES NC(=N)c1ccc(\C=C2\CCCC\C(=C\c3ccc(cc3)C(N)=N)C2=O)cc1 Show InChI InChI=1S/C23H24N4O/c24-22(25)17-9-5-15(6-10-17)13-19-3-1-2-4-20(21(19)28)14-16-7-11-18(12-8-16)23(26)27/h5-14H,1-4H2,(H3,24,25)(H3,26,27)/b19-13-,20-14- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 0.660 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
In vitro inhibition of human coagulation factor Xa (Xa) in a purified enzyme system. |
J Med Chem 42: 5415-25 (2000)
BindingDB Entry DOI: 10.7270/Q2TT4Q5S |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM17277

((Z,Z)BABCH | 4-{[(1Z,3Z)-3-[(4-carbamimidoylphenyl...)Show SMILES NC(=N)c1ccc(\C=C2\CCCC\C(=C\c3ccc(cc3)C(N)=N)C2=O)cc1 Show InChI InChI=1S/C23H24N4O/c24-22(25)17-9-5-15(6-10-17)13-19-3-1-2-4-20(21(19)28)14-16-7-11-18(12-8-16)23(26)27/h5-14H,1-4H2,(H3,24,25)(H3,26,27)/b19-13-,20-14- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.660 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Compound (isomer) was tested in the absence of light for inhibitory activity against Human Coagulation factor X |
J Med Chem 41: 3551-6 (1998)
Article DOI: 10.1021/jm980281+
BindingDB Entry DOI: 10.7270/Q2QJ7GFX |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM17277

((Z,Z)BABCH | 4-{[(1Z,3Z)-3-[(4-carbamimidoylphenyl...)Show SMILES NC(=N)c1ccc(\C=C2\CCCC\C(=C\c3ccc(cc3)C(N)=N)C2=O)cc1 Show InChI InChI=1S/C23H24N4O/c24-22(25)17-9-5-15(6-10-17)13-19-3-1-2-4-20(21(19)28)14-16-7-11-18(12-8-16)23(26)27/h5-14H,1-4H2,(H3,24,25)(H3,26,27)/b19-13-,20-14- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 0.660 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Inhibitory activity against human Coagulation factor X |
Bioorg Med Chem Lett 10: 957-61 (2000)
BindingDB Entry DOI: 10.7270/Q2MG7NRD |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM17277

((Z,Z)BABCH | 4-{[(1Z,3Z)-3-[(4-carbamimidoylphenyl...)Show SMILES NC(=N)c1ccc(\C=C2\CCCC\C(=C\c3ccc(cc3)C(N)=N)C2=O)cc1 Show InChI InChI=1S/C23H24N4O/c24-22(25)17-9-5-15(6-10-17)13-19-3-1-2-4-20(21(19)28)14-16-7-11-18(12-8-16)23(26)27/h5-14H,1-4H2,(H3,24,25)(H3,26,27)/b19-13-,20-14- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.660 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Inhibitory potency was measured against human coagulation factor X |
J Med Chem 41: 3557-62 (1998)
Article DOI: 10.1021/jm980280h
BindingDB Entry DOI: 10.7270/Q2KS6QPR |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM17277

((Z,Z)BABCH | 4-{[(1Z,3Z)-3-[(4-carbamimidoylphenyl...)Show SMILES NC(=N)c1ccc(\C=C2\CCCC\C(=C\c3ccc(cc3)C(N)=N)C2=O)cc1 Show InChI InChI=1S/C23H24N4O/c24-22(25)17-9-5-15(6-10-17)13-19-3-1-2-4-20(21(19)28)14-16-7-11-18(12-8-16)23(26)27/h5-14H,1-4H2,(H3,24,25)(H3,26,27)/b19-13-,20-14- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Binding affinity of the compound was evaluated against Coagulation factor X |
Bioorg Med Chem Lett 8: 1877-82 (1999)
BindingDB Entry DOI: 10.7270/Q2222SX9 |
More data for this
Ligand-Target Pair | |
Trypsin
(Bos taurus (bovine)) | BDBM17277

((Z,Z)BABCH | 4-{[(1Z,3Z)-3-[(4-carbamimidoylphenyl...)Show SMILES NC(=N)c1ccc(\C=C2\CCCC\C(=C\c3ccc(cc3)C(N)=N)C2=O)cc1 Show InChI InChI=1S/C23H24N4O/c24-22(25)17-9-5-15(6-10-17)13-19-3-1-2-4-20(21(19)28)14-16-7-11-18(12-8-16)23(26)27/h5-14H,1-4H2,(H3,24,25)(H3,26,27)/b19-13-,20-14- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Compound was tested for inhibitory activity against bovine trypsin |
Bioorg Med Chem Lett 8: 2235-40 (1999)
BindingDB Entry DOI: 10.7270/Q2G44PGD |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Trypsin
(Bos taurus (bovine)) | BDBM17277

((Z,Z)BABCH | 4-{[(1Z,3Z)-3-[(4-carbamimidoylphenyl...)Show SMILES NC(=N)c1ccc(\C=C2\CCCC\C(=C\c3ccc(cc3)C(N)=N)C2=O)cc1 Show InChI InChI=1S/C23H24N4O/c24-22(25)17-9-5-15(6-10-17)13-19-3-1-2-4-20(21(19)28)14-16-7-11-18(12-8-16)23(26)27/h5-14H,1-4H2,(H3,24,25)(H3,26,27)/b19-13-,20-14- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 33 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Compound (isomer) was tested in the absence of light for inhibitory activity against trypsin. |
J Med Chem 41: 3551-6 (1998)
Article DOI: 10.1021/jm980281+
BindingDB Entry DOI: 10.7270/Q2QJ7GFX |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Trypsin
(Bos taurus (bovine)) | BDBM17277

((Z,Z)BABCH | 4-{[(1Z,3Z)-3-[(4-carbamimidoylphenyl...)Show SMILES NC(=N)c1ccc(\C=C2\CCCC\C(=C\c3ccc(cc3)C(N)=N)C2=O)cc1 Show InChI InChI=1S/C23H24N4O/c24-22(25)17-9-5-15(6-10-17)13-19-3-1-2-4-20(21(19)28)14-16-7-11-18(12-8-16)23(26)27/h5-14H,1-4H2,(H3,24,25)(H3,26,27)/b19-13-,20-14- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 33 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Inhibitory potency was measured against bovine trypsin. |
J Med Chem 41: 3557-62 (1998)
Article DOI: 10.1021/jm980280h
BindingDB Entry DOI: 10.7270/Q2KS6QPR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Trypsin
(Bos taurus (bovine)) | BDBM17277

((Z,Z)BABCH | 4-{[(1Z,3Z)-3-[(4-carbamimidoylphenyl...)Show SMILES NC(=N)c1ccc(\C=C2\CCCC\C(=C\c3ccc(cc3)C(N)=N)C2=O)cc1 Show InChI InChI=1S/C23H24N4O/c24-22(25)17-9-5-15(6-10-17)13-19-3-1-2-4-20(21(19)28)14-16-7-11-18(12-8-16)23(26)27/h5-14H,1-4H2,(H3,24,25)(H3,26,27)/b19-13-,20-14- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 33 | -10.1 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
Berlex
| Assay Description
The enzyme activities were determined kinetically as the initial rate of cleavage of a peptide p-nitroanilide. Km for enzyme and substrate was determ... |
Acta Crystallogr D Biol Crystallogr 55: 1395-404 (1999)
Article DOI: 10.1107/s0907444999007350
BindingDB Entry DOI: 10.7270/Q2H1308B |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Trypsin
(Bos taurus (bovine)) | BDBM17277

((Z,Z)BABCH | 4-{[(1Z,3Z)-3-[(4-carbamimidoylphenyl...)Show SMILES NC(=N)c1ccc(\C=C2\CCCC\C(=C\c3ccc(cc3)C(N)=N)C2=O)cc1 Show InChI InChI=1S/C23H24N4O/c24-22(25)17-9-5-15(6-10-17)13-19-3-1-2-4-20(21(19)28)14-16-7-11-18(12-8-16)23(26)27/h5-14H,1-4H2,(H3,24,25)(H3,26,27)/b19-13-,20-14- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
PubMed
| 33 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
In vitro inhibition of bovine trypsin(Trp). |
J Med Chem 42: 5415-25 (2000)
BindingDB Entry DOI: 10.7270/Q2TT4Q5S |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prothrombin
(Homo sapiens (Human)) | BDBM17277

((Z,Z)BABCH | 4-{[(1Z,3Z)-3-[(4-carbamimidoylphenyl...)Show SMILES NC(=N)c1ccc(\C=C2\CCCC\C(=C\c3ccc(cc3)C(N)=N)C2=O)cc1 Show InChI InChI=1S/C23H24N4O/c24-22(25)17-9-5-15(6-10-17)13-19-3-1-2-4-20(21(19)28)14-16-7-11-18(12-8-16)23(26)27/h5-14H,1-4H2,(H3,24,25)(H3,26,27)/b19-13-,20-14- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Compound was tested for inhibitory activity against human thrombin |
Bioorg Med Chem Lett 8: 2235-40 (1999)
BindingDB Entry DOI: 10.7270/Q2G44PGD |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM17277

((Z,Z)BABCH | 4-{[(1Z,3Z)-3-[(4-carbamimidoylphenyl...)Show SMILES NC(=N)c1ccc(\C=C2\CCCC\C(=C\c3ccc(cc3)C(N)=N)C2=O)cc1 Show InChI InChI=1S/C23H24N4O/c24-22(25)17-9-5-15(6-10-17)13-19-3-1-2-4-20(21(19)28)14-16-7-11-18(12-8-16)23(26)27/h5-14H,1-4H2,(H3,24,25)(H3,26,27)/b19-13-,20-14- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Compound (isomer) was tested in the absence of light for inhibitory activity against thrombin. |
J Med Chem 41: 3551-6 (1998)
Article DOI: 10.1021/jm980281+
BindingDB Entry DOI: 10.7270/Q2QJ7GFX |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM17277

((Z,Z)BABCH | 4-{[(1Z,3Z)-3-[(4-carbamimidoylphenyl...)Show SMILES NC(=N)c1ccc(\C=C2\CCCC\C(=C\c3ccc(cc3)C(N)=N)C2=O)cc1 Show InChI InChI=1S/C23H24N4O/c24-22(25)17-9-5-15(6-10-17)13-19-3-1-2-4-20(21(19)28)14-16-7-11-18(12-8-16)23(26)27/h5-14H,1-4H2,(H3,24,25)(H3,26,27)/b19-13-,20-14- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
In vitro inhibition of human thrombin(FIIa). |
J Med Chem 42: 5415-25 (2000)
BindingDB Entry DOI: 10.7270/Q2TT4Q5S |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM17277

((Z,Z)BABCH | 4-{[(1Z,3Z)-3-[(4-carbamimidoylphenyl...)Show SMILES NC(=N)c1ccc(\C=C2\CCCC\C(=C\c3ccc(cc3)C(N)=N)C2=O)cc1 Show InChI InChI=1S/C23H24N4O/c24-22(25)17-9-5-15(6-10-17)13-19-3-1-2-4-20(21(19)28)14-16-7-11-18(12-8-16)23(26)27/h5-14H,1-4H2,(H3,24,25)(H3,26,27)/b19-13-,20-14- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Inhibitory potency was measured against human thrombin |
J Med Chem 41: 3557-62 (1998)
Article DOI: 10.1021/jm980280h
BindingDB Entry DOI: 10.7270/Q2KS6QPR |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data