

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM17678 2-aminobenzimidazole, 3::4-[(2-{[4-chloro-3-(trifluoromethyl)phenyl]amino}-1H-1,3-benzodiazol-5-yl)oxy]-N-methylpyridine-2-carboxamide::JMC517049 Compound 3
SMILES: CNC(=O)c1cc(Oc2ccc3nc(Nc4ccc(Cl)c(c4)C(F)(F)F)[nH]c3c2)ccn1
InChI Key: InChIKey=ZJLSMLDOCGOURY-UHFFFAOYSA-N
PDB links: 1 PDB ID matches this monomer. 1 PDB ID contains inhibitors having a similarity of 90% to this monomer.
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Vascular endothelial growth factor receptor 2 (Homo sapiens (Human)) | BDBM17678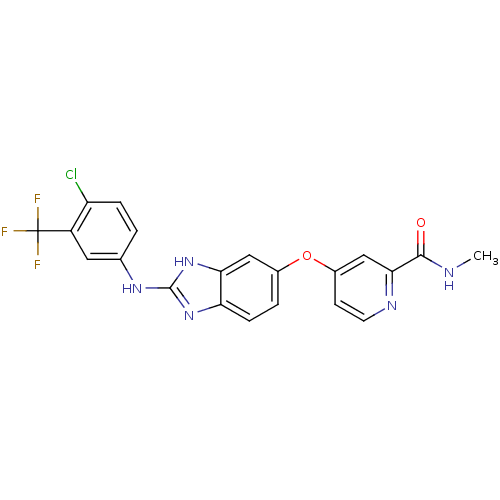 (2-aminobenzimidazole, 3 | 4-[(2-{[4-chloro-3-(trif...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 9 | -10.9 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
Amgen | Assay Description The assay involves the phosphorylation of a biotinylated substrate and the detection of this phosphorylation after the addition of a streptavidin-all... | J Med Chem 50: 4351-4373 (2007) Article DOI: 10.1021/jm070034i BindingDB Entry DOI: 10.7270/Q21V5C74 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| B-RAF V600E (Homo sapiens (Human)) | BDBM17678 (2-aminobenzimidazole, 3 | 4-[(2-{[4-chloro-3-(trif...) | PDB GoogleScholar AffyNet | Purchase DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 280 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis | Assay Description To measure compound inhibitor effects on the ability of Raf enzymes to phosphorylate the Mek substrate, a capture ELISA utilizing streptavidin-coated... | J Med Chem 51: 7049-52 (2008) Article DOI: 10.1021/jm801050k BindingDB Entry DOI: 10.7270/Q2B56H1C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| RAF proto-oncogene serine/threonine-protein kinase (Homo sapiens (Human)) | BDBM17678 (2-aminobenzimidazole, 3 | 4-[(2-{[4-chloro-3-(trif...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 76 | n/a | n/a | n/a | n/a | 7.8 | 22 |
Novartis | Assay Description To measure compound inhibitor effects on the ability of Raf enzymes to phosphorylate the Mek substrate, a capture ELISA utilizing streptavidin-coated... | J Med Chem 51: 7049-52 (2008) Article DOI: 10.1021/jm801050k BindingDB Entry DOI: 10.7270/Q2B56H1C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||