 Found 5 hits for monomerid = 18375
Found 5 hits for monomerid = 18375 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Bile salt export pump (BSEP)
(Homo sapiens (Human)) | BDBM18375
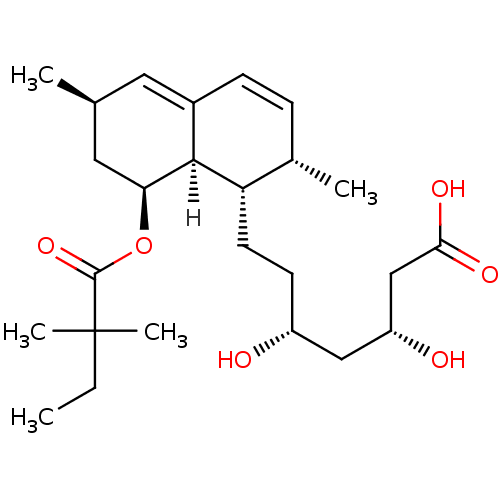
((3R,5R)-7-[(1S,2S,6R,8S,8aR)-8-[(2,2-dimethylbutan...)Show SMILES [H][C@]12[C@H](C[C@@H](C)C=C1C=C[C@H](C)[C@@H]2CC[C@@H](O)C[C@@H](O)CC(O)=O)OC(=O)C(C)(C)CC |r,c:6,9| Show InChI InChI=1S/C25H40O6/c1-6-25(4,5)24(30)31-21-12-15(2)11-17-8-7-16(3)20(23(17)21)10-9-18(26)13-19(27)14-22(28)29/h7-8,11,15-16,18-21,23,26-27H,6,9-10,12-14H2,1-5H3,(H,28,29)/t15-,16-,18+,19+,20-,21-,23-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tokyo
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 314: 876-82 (2005)
Article DOI: 10.1124/jpet.105.084830
BindingDB Entry DOI: 10.7270/Q23X8573 |
More data for this
Ligand-Target Pair | |
Bile Salt Export Pump, BSEP
(Rattus norvegicus) | BDBM18375

((3R,5R)-7-[(1S,2S,6R,8S,8aR)-8-[(2,2-dimethylbutan...)Show SMILES [H][C@]12[C@H](C[C@@H](C)C=C1C=C[C@H](C)[C@@H]2CC[C@@H](O)C[C@@H](O)CC(O)=O)OC(=O)C(C)(C)CC |r,c:6,9| Show InChI InChI=1S/C25H40O6/c1-6-25(4,5)24(30)31-21-12-15(2)11-17-8-7-16(3)20(23(17)21)10-9-18(26)13-19(27)14-22(28)29/h7-8,11,15-16,18-21,23,26-27H,6,9-10,12-14H2,1-5H3,(H,28,29)/t15-,16-,18+,19+,20-,21-,23-/m0/s1 | KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| Purchase
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tokyo
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 314: 876-82 (2005)
Article DOI: 10.1124/jpet.105.084830
BindingDB Entry DOI: 10.7270/Q23X8573 |
More data for this
Ligand-Target Pair | |
Solute carrier organic anion transporter family member 1B1 (OATP1B1)
(Homo sapiens (Human)) | BDBM18375

((3R,5R)-7-[(1S,2S,6R,8S,8aR)-8-[(2,2-dimethylbutan...)Show SMILES [H][C@]12[C@H](C[C@@H](C)C=C1C=C[C@H](C)[C@@H]2CC[C@@H](O)C[C@@H](O)CC(O)=O)OC(=O)C(C)(C)CC |r,c:6,9| Show InChI InChI=1S/C25H40O6/c1-6-25(4,5)24(30)31-21-12-15(2)11-17-8-7-16(3)20(23(17)21)10-9-18(26)13-19(27)14-22(28)29/h7-8,11,15-16,18-21,23,26-27H,6,9-10,12-14H2,1-5H3,(H,28,29)/t15-,16-,18+,19+,20-,21-,23-/m0/s1 | UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
TP_TRANSPORTER: inhibition of estradiol-17beta-glucuronide uptake(estradiol-17beta-glucuronide:0.02uM) in OATP1B1-expressing HEK293 cells |
Drug Metab Dispos 33: 537-46 (2005)
Article DOI: 10.1124/dmd.104.002477
BindingDB Entry DOI: 10.7270/Q2SB4712 |
More data for this
Ligand-Target Pair | |
HMG-CoA reductase
(Rattus norvegicus (rat)) | BDBM18375

((3R,5R)-7-[(1S,2S,6R,8S,8aR)-8-[(2,2-dimethylbutan...)Show SMILES [H][C@]12[C@H](C[C@@H](C)C=C1C=C[C@H](C)[C@@H]2CC[C@@H](O)C[C@@H](O)CC(O)=O)OC(=O)C(C)(C)CC |r,c:6,9| Show InChI InChI=1S/C25H40O6/c1-6-25(4,5)24(30)31-21-12-15(2)11-17-8-7-16(3)20(23(17)21)10-9-18(26)13-19(27)14-22(28)29/h7-8,11,15-16,18-21,23,26-27H,6,9-10,12-14H2,1-5H3,(H,28,29)/t15-,16-,18+,19+,20-,21-,23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 4.30 | n/a | 6.20 | n/a | n/a | 7.0 | 37 |
Bristol-Myers Squibb Company
| Assay Description
Enzyme Assay for HMG-CoA reductase was based on the conversion of isotopically labeled HMG-CoA to mevalonic acid using rat liver microsomes as enzyme... |
J Med Chem 51: 2722-33 (2008)
Article DOI: 10.1021/jm800001n
BindingDB Entry DOI: 10.7270/Q2FT8JB2 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
HMG-CoA reductase
(Rattus norvegicus (rat)) | BDBM18375

((3R,5R)-7-[(1S,2S,6R,8S,8aR)-8-[(2,2-dimethylbutan...)Show SMILES [H][C@]12[C@H](C[C@@H](C)C=C1C=C[C@H](C)[C@@H]2CC[C@@H](O)C[C@@H](O)CC(O)=O)OC(=O)C(C)(C)CC |r,c:6,9| Show InChI InChI=1S/C25H40O6/c1-6-25(4,5)24(30)31-21-12-15(2)11-17-8-7-16(3)20(23(17)21)10-9-18(26)13-19(27)14-22(28)29/h7-8,11,15-16,18-21,23,26-27H,6,9-10,12-14H2,1-5H3,(H,28,29)/t15-,16-,18+,19+,20-,21-,23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | 7.2 | 37 |
Pfizer
| Assay Description
Assay for HMG-CoA reductase was based on the conversion of isotopically labeled HMG-CoA to mevalonic acid using rat liver microsomes as enzyme source... |
Bioorg Med Chem 15: 5576-89 (2007)
Article DOI: 10.1016/j.bmc.2007.05.031
BindingDB Entry DOI: 10.7270/Q23J3B7G |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data