 Found 12 hits for monomerid = 2
Found 12 hits for monomerid = 2 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
adrenergic Alpha2
(RAT) | BDBM2
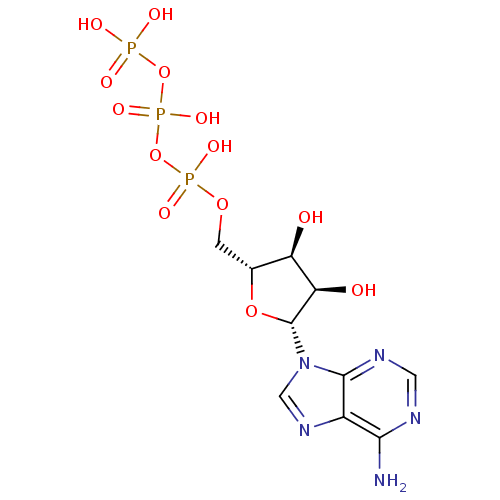
(({[({[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C10H16N5O13P3/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(17)6(16)4(26-10)1-25-30(21,22)28-31(23,24)27-29(18,19)20/h2-4,6-7,10,16-17H,1H2,(H,21,22)(H,23,24)(H2,11,12,13)(H2,18,19,20)/t4-,6-,7-,10-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 3.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nebraska
Curated by PDSP Ki Database
| |
J Neurosci Methods 105: 159-66 (2001)
Article DOI: 10.1016/s0165-0270(00)00358-7
BindingDB Entry DOI: 10.7270/Q2NS0SFS |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 3
(Homo sapiens (Human)) | BDBM2

(({[({[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C10H16N5O13P3/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(17)6(16)4(26-10)1-25-30(21,22)28-31(23,24)27-29(18,19)20/h2-4,6-7,10,16-17H,1H2,(H,21,22)(H,23,24)(H2,11,12,13)(H2,18,19,20)/t4-,6-,7-,10-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 14.6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 407-16 (2004)
Article DOI: 10.1124/jpet.103.064907
BindingDB Entry DOI: 10.7270/Q2SQ8XZR |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 2
(Homo sapiens (Human)) | BDBM2

(({[({[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C10H16N5O13P3/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(17)6(16)4(26-10)1-25-30(21,22)28-31(23,24)27-29(18,19)20/h2-4,6-7,10,16-17H,1H2,(H,21,22)(H,23,24)(H2,11,12,13)(H2,18,19,20)/t4-,6-,7-,10-/m1/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 14.7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 407-16 (2004)
Article DOI: 10.1124/jpet.103.064907
BindingDB Entry DOI: 10.7270/Q2SQ8XZR |
More data for this
Ligand-Target Pair | |
Cholecystokinin
(RAT) | BDBM2

(({[({[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C10H16N5O13P3/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(17)6(16)4(26-10)1-25-30(21,22)28-31(23,24)27-29(18,19)20/h2-4,6-7,10,16-17H,1H2,(H,21,22)(H,23,24)(H2,11,12,13)(H2,18,19,20)/t4-,6-,7-,10-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche & Co., Ltd.
Curated by PDSP Ki Database
| |
J Neurosci 4: 1021-33 (1984)
BindingDB Entry DOI: 10.7270/Q2P55M0W |
More data for this
Ligand-Target Pair | |
Cholecystokinin
(RAT) | BDBM2

(({[({[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C10H16N5O13P3/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(17)6(16)4(26-10)1-25-30(21,22)28-31(23,24)27-29(18,19)20/h2-4,6-7,10,16-17H,1H2,(H,21,22)(H,23,24)(H2,11,12,13)(H2,18,19,20)/t4-,6-,7-,10-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche & Co., Ltd.
Curated by PDSP Ki Database
| |
J Neurosci 4: 1021-33 (1984)
BindingDB Entry DOI: 10.7270/Q2P55M0W |
More data for this
Ligand-Target Pair | |
tRNA synthetase (GlyRS)
(Bombyx mori) | BDBM2

(({[({[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C10H16N5O13P3/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(17)6(16)4(26-10)1-25-30(21,22)28-31(23,24)27-29(18,19)20/h2-4,6-7,10,16-17H,1H2,(H,21,22)(H,23,24)(H2,11,12,13)(H2,18,19,20)/t4-,6-,7-,10-/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 1.30E+4 | -6.66 | 1.00E+5 | n/a | n/a | n/a | n/a | 7.5 | 25 |
Medical College of Ohio
| Assay Description
Aminoacyl-tRNA synthetase assays were measuring the incorporation of [14C] amino acid into tRNA. |
Biochemistry 42: 5333-40 (2003)
Article DOI: 10.1021/bi030031h
BindingDB Entry DOI: 10.7270/Q2HQ3X6H |
More data for this
Ligand-Target Pair | |
Purinergic receptor P2Y1
(Homo sapiens (Human)) | BDBM2

(({[({[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C10H16N5O13P3/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(17)6(16)4(26-10)1-25-30(21,22)28-31(23,24)27-29(18,19)20/h2-4,6-7,10,16-17H,1H2,(H,21,22)(H,23,24)(H2,11,12,13)(H2,18,19,20)/t4-,6-,7-,10-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 150 | n/a | n/a | n/a | n/a |
Bar-Ilan University
Curated by ChEMBL
| Assay Description
Agonist activity at GFP tagged-human P2Y1 receptor expressed in human 1321N1 cells assessed as elevation in calcium level after 30 mins by fluorescen... |
J Med Chem 53: 8485-97 (2010)
Article DOI: 10.1021/jm100597c
BindingDB Entry DOI: 10.7270/Q22V2GF7 |
More data for this
Ligand-Target Pair | |
Purinergic receptor P2Y11
(Homo sapiens (Human)) | BDBM2

(({[({[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C10H16N5O13P3/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(17)6(16)4(26-10)1-25-30(21,22)28-31(23,24)27-29(18,19)20/h2-4,6-7,10,16-17H,1H2,(H,21,22)(H,23,24)(H2,11,12,13)(H2,18,19,20)/t4-,6-,7-,10-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 3.30E+3 | n/a | n/a | n/a | n/a |
Bar-Ilan University
Curated by ChEMBL
| Assay Description
Agonist activity at GFP tagged-human P2Y11 receptor expressed in human 1321N1 cells assessed as elevation in calcium level after 30 mins by fluoresce... |
J Med Chem 53: 8485-97 (2010)
Article DOI: 10.1021/jm100597c
BindingDB Entry DOI: 10.7270/Q22V2GF7 |
More data for this
Ligand-Target Pair | |
Purinergic receptor P2Y1
(Homo sapiens (Human)) | BDBM2

(({[({[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C10H16N5O13P3/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(17)6(16)4(26-10)1-25-30(21,22)28-31(23,24)27-29(18,19)20/h2-4,6-7,10,16-17H,1H2,(H,21,22)(H,23,24)(H2,11,12,13)(H2,18,19,20)/t4-,6-,7-,10-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 200 | n/a | n/a | n/a | n/a |
Bar-Ilan University
Curated by ChEMBL
| Assay Description
Agonist activity at P2Y1 receptor expressed in human HEK293 cells |
J Med Chem 53: 2472-81 (2010)
Article DOI: 10.1021/jm901621h
BindingDB Entry DOI: 10.7270/Q2DN4565 |
More data for this
Ligand-Target Pair | |
P2Y purinoceptor 1
(Rattus norvegicus) | BDBM2

(({[({[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C10H16N5O13P3/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(17)6(16)4(26-10)1-25-30(21,22)28-31(23,24)27-29(18,19)20/h2-4,6-7,10,16-17H,1H2,(H,21,22)(H,23,24)(H2,11,12,13)(H2,18,19,20)/t4-,6-,7-,10-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 590 | n/a | n/a | n/a | n/a |
Bar-Ilan University
Curated by ChEMBL
| Assay Description
Concentration required for calcium mobilization at rat purinergic 2Y1 receptor expressed in HEK 293 cells |
J Med Chem 47: 4405-16 (2004)
Article DOI: 10.1021/jm049771u
BindingDB Entry DOI: 10.7270/Q2PC334S |
More data for this
Ligand-Target Pair | |
Heat Shock 70kDa Protein 1
(Homo sapiens (Human)) | BDBM2

(({[({[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C10H16N5O13P3/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(17)6(16)4(26-10)1-25-30(21,22)28-31(23,24)27-29(18,19)20/h2-4,6-7,10,16-17H,1H2,(H,21,22)(H,23,24)(H2,11,12,13)(H2,18,19,20)/t4-,6-,7-,10-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 500 | n/a | n/a | n/a | n/a | 7.4 | 23 |
Vernalis (R&D) Ltd.
| Assay Description
A fluorescein-labeled ATP-based probe was utilized, which binds to the GST fused ATPase domain (amino acids 3-383) of HSP70, producing an increase in... |
J Med Chem 52: 1510-3 (2009)
Article DOI: 10.1021/jm801627a
BindingDB Entry DOI: 10.7270/Q29S1PDC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Purinergic receptor P2Y2
(Homo sapiens (Human)) | BDBM2

(({[({[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C10H16N5O13P3/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(17)6(16)4(26-10)1-25-30(21,22)28-31(23,24)27-29(18,19)20/h2-4,6-7,10,16-17H,1H2,(H,21,22)(H,23,24)(H2,11,12,13)(H2,18,19,20)/t4-,6-,7-,10-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 85 | n/a | n/a | n/a | n/a |
Ghent University
Curated by ChEMBL
| Assay Description
Agonist activity at human P2Y2 receptor expressed in human 1321N1 cells coexpressing phospholipase C-activating G protein assessed as inositol phosph... |
Bioorg Med Chem Lett 19: 3002-5 (2009)
Article DOI: 10.1016/j.bmcl.2009.04.027
BindingDB Entry DOI: 10.7270/Q20K28GT |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data




