

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM21139 (3S)-3-[(2S)-2-[(2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-(4-hydroxyphenyl)propanamido]hexanamido]acetamido}-3-(1H-indol-3-yl)propanamido]hexanamido]-3-{[(1S)-1-carbamoyl-2-phenylethyl]carbamoyl}propanoic acid::CCK-Opioid Peptide, 9::Tyr-D-Nle-Gly-Trp-Nle-Asp-Phe-NH2
SMILES: CCCC[C@@H](NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCCC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O
InChI Key: InChIKey=YVOYRMXNVINXII-ULSNPNCNSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM21139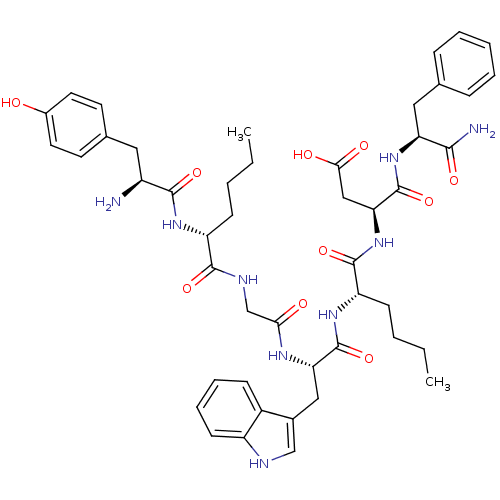 ((3S)-3-[(2S)-2-[(2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 2.60 | -11.7 | n/a | n/a | 4.5 | n/a | n/a | 7.4 | 25 |
University of Arizona at Tucson | Assay Description Log IC50 values for each test compound were determined from nonlinear regression analysis of data collected from two independent experiments performe... | J Med Chem 49: 2868-75 (2006) Article DOI: 10.1021/jm050921q BindingDB Entry DOI: 10.7270/Q24Q7S99 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholecystokinin receptor (Homo sapiens (Human)) | BDBM21139 ((3S)-3-[(2S)-2-[(2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 9.60 | n/a | n/a | n/a | 2.50E+3 | n/a | n/a | n/a | n/a |
University of Arizona at Tucson | Assay Description Log IC50 values for each test compound were determined from nonlinear regression analysis of data collected from two independent experiments performe... | J Med Chem 49: 2868-75 (2006) Article DOI: 10.1021/jm050921q BindingDB Entry DOI: 10.7270/Q24Q7S99 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholecystokinin receptor (Homo sapiens (Human)) | BDBM21139 ((3S)-3-[(2S)-2-[(2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 15 | n/a | n/a | n/a | 1.90E+3 | n/a | n/a | n/a | n/a |
University of Arizona at Tucson | Assay Description Log IC50 values for each test compound were determined from nonlinear regression analysis of data collected from two independent experiments performe... | J Med Chem 49: 2868-75 (2006) Article DOI: 10.1021/jm050921q BindingDB Entry DOI: 10.7270/Q24Q7S99 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM21139 ((3S)-3-[(2S)-2-[(2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 26 | -10.3 | n/a | n/a | 0.460 | n/a | n/a | 7.4 | 25 |
University of Arizona at Tucson | Assay Description Log IC50 values for each test compound were determined from nonlinear regression analysis of data collected from two independent experiments performe... | J Med Chem 49: 2868-75 (2006) Article DOI: 10.1021/jm050921q BindingDB Entry DOI: 10.7270/Q24Q7S99 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||