

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM221048 US9284322, varenicline::US9303017, Varenicline
SMILES: C1[C@H]2CNC[C@@H]1c1cc3nccnc3cc21
InChI Key: InChIKey=JQSHBVHOMNKWFT-DTORHVGOSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Neuronal acetylcholine receptor Alpha-4/Beta-2 (Homo sapiens (Human)) | BDBM221048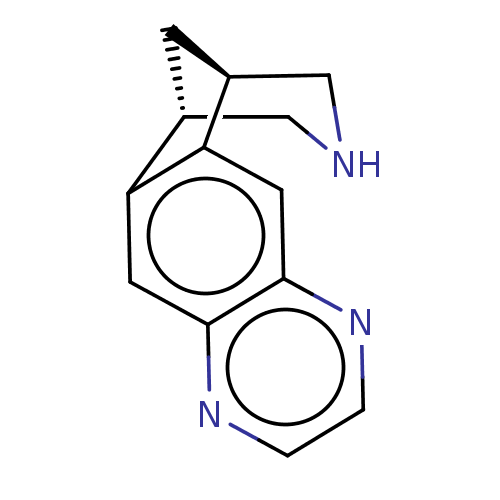 (US9284322, varenicline | US9303017, Varenicline) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid PDB UniChem | PDB PubMed | 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]cytisine from human alpha4beta2 nAChR expressed in CHOK1 cell membrane by microbeta scintillation counting method | J Med Chem 63: 2833-2853 (2020) | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Neuronal acetylcholine receptor Alpha-4/Beta-2 (Homo sapiens (Human)) | BDBM221048 (US9284322, varenicline | US9303017, Varenicline) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid PDB UniChem | PDB US Patent | 0.120 | -12.6 | n/a | n/a | n/a | n/a | n/a | 7.4 | 4 |
Georgetown University; Duke University US Patent | Assay Description Briefly, cultured cells at >80% confluence were removed from their flasks (80 cm^2) with a disposable cell scraper and placed in 10 mL of 50 mM Tris.... | US Patent US9303017 (2016) BindingDB Entry DOI: 10.7270/Q25X27SF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| nAChR alpha4 beta2 subtype (Rattus norvegicus (Rat)) | BDBM221048 (US9284322, varenicline | US9303017, Varenicline) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid PDB UniChem | PDB US Patent | 0.120 | -13.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Research Triangle Institute US Patent | Assay Description Adult male rat cerebral cortices (Pelfreeze Biological, Rogers, Ark.) were homogenized in 39 volumes of ice-cold 50 mM Tris buffer (pH 7.4 at 4� C.) ... | US Patent US9284322 (2016) BindingDB Entry DOI: 10.7270/Q2QV3KCV | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Neuronal acetylcholine receptor protein alpha-2/beta-2 subunit (Rattus norvegicus (Rat)) | BDBM221048 (US9284322, varenicline | US9303017, Varenicline) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid PDB UniChem | PDB US Patent | 0.480 | -11.8 | n/a | n/a | n/a | n/a | n/a | 7.4 | 4 |
Georgetown University; Duke University US Patent | Assay Description Briefly, cultured cells at >80% confluence were removed from their flasks (80 cm^2) with a disposable cell scraper and placed in 10 mL of 50 mM Tris.... | US Patent US9303017 (2016) BindingDB Entry DOI: 10.7270/Q25X27SF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Neuronal acetylcholine receptor Alpha-3/Beta-2 (Rattus norvegicus (Rat)) | BDBM221048 (US9284322, varenicline | US9303017, Varenicline) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid PDB UniChem | PDB US Patent | 2.5 | -10.9 | n/a | n/a | n/a | n/a | n/a | 7.4 | 4 |
Georgetown University; Duke University US Patent | Assay Description Briefly, cultured cells at >80% confluence were removed from their flasks (80 cm^2) with a disposable cell scraper and placed in 10 mL of 50 mM Tris.... | US Patent US9303017 (2016) BindingDB Entry DOI: 10.7270/Q25X27SF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Neuronal acetylcholine receptor protein alpha-4/beta-4 subunit (Rattus norvegicus (Rat)) | BDBM221048 (US9284322, varenicline | US9303017, Varenicline) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid PDB UniChem | PDB US Patent | 28 | -9.57 | n/a | n/a | n/a | n/a | n/a | 7.4 | 4 |
Georgetown University; Duke University US Patent | Assay Description Briefly, cultured cells at >80% confluence were removed from their flasks (80 cm^2) with a disposable cell scraper and placed in 10 mL of 50 mM Tris.... | US Patent US9303017 (2016) BindingDB Entry DOI: 10.7270/Q25X27SF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Neuronal acetylcholine receptor (Rattus norvegicus (Rat)) | BDBM221048 (US9284322, varenicline | US9303017, Varenicline) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid PDB UniChem | US Patent | 32.5 | n/a | n/a | n/a | n/a | n/a | n/a | 7.4 | n/a |
Research Triangle Institute US Patent | Assay Description Adult male rat cerebral cortices (Pel-Freez Biologicals, Rogers, Ark.) were homogenized (polytron) in 39 volumes of ice-cold 50 mM Tris buffer (assay... | US Patent US9284322 (2016) BindingDB Entry DOI: 10.7270/Q2QV3KCV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor (Rattus norvegicus (Rat)) | BDBM221048 (US9284322, varenicline | US9303017, Varenicline) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid PDB UniChem | US Patent | 37 | -9.42 | n/a | n/a | n/a | n/a | n/a | 7.4 | 4 |
Georgetown University; Duke University US Patent | Assay Description Briefly, cultured cells at >80% confluence were removed from their flasks (80 cm^2) with a disposable cell scraper and placed in 10 mL of 50 mM Tris.... | US Patent US9303017 (2016) BindingDB Entry DOI: 10.7270/Q25X27SF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor; alpha3/beta4 (Homo sapiens (Human)) | BDBM221048 (US9284322, varenicline | US9303017, Varenicline) | PDB KEGG UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid PDB UniChem | PubMed | 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]epibatidine from human alpha3beta4 nAChR expressed in IMR32 cell membrane by microbeta scintillation counting method | J Med Chem 63: 2833-2853 (2020) | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor Alpha-2/Beta-4 (Rattus norvegicus (Rat)) | BDBM221048 (US9284322, varenicline | US9303017, Varenicline) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid PDB UniChem | US Patent | 94 | -8.90 | n/a | n/a | n/a | n/a | n/a | 7.4 | 4 |
Georgetown University; Duke University US Patent | Assay Description Briefly, cultured cells at >80% confluence were removed from their flasks (80 cm^2) with a disposable cell scraper and placed in 10 mL of 50 mM Tris.... | US Patent US9303017 (2016) BindingDB Entry DOI: 10.7270/Q25X27SF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor Alpha-3/Beta-4 (Rattus norvegicus (Rat)) | BDBM221048 (US9284322, varenicline | US9303017, Varenicline) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid PDB UniChem | US Patent | 390 | -8.12 | n/a | n/a | n/a | n/a | n/a | 7.4 | 4 |
Georgetown University; Duke University US Patent | Assay Description Briefly, cultured cells at >80% confluence were removed from their flasks (80 cm^2) with a disposable cell scraper and placed in 10 mL of 50 mM Tris.... | US Patent US9303017 (2016) BindingDB Entry DOI: 10.7270/Q25X27SF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor Alpha-3/Beta-4 (Rattus norvegicus (Rat)) | BDBM221048 (US9284322, varenicline | US9303017, Varenicline) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid PDB UniChem | US Patent | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Georgetown University; Duke University US Patent | Assay Description IC50(10′): The functional properties of the ligands were determined by 86Rb+ efflux assays in cells expressing α3β4 and α4β... | US Patent US9303017 (2016) BindingDB Entry DOI: 10.7270/Q25X27SF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor Alpha-4/Beta-2 (Homo sapiens (Human)) | BDBM221048 (US9284322, varenicline | US9303017, Varenicline) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid PDB UniChem | PDB US Patent | n/a | n/a | 94 | n/a | n/a | n/a | n/a | n/a | n/a |
Georgetown University; Duke University US Patent | Assay Description IC50(10′): The functional properties of the ligands were determined by 86Rb+ efflux assays in cells expressing α3β4 and α4β... | US Patent US9303017 (2016) BindingDB Entry DOI: 10.7270/Q25X27SF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Neuronal acetylcholine receptor Alpha-4/Beta-2 (Homo sapiens (Human)) | BDBM221048 (US9284322, varenicline | US9303017, Varenicline) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid PDB UniChem | PDB US Patent | n/a | n/a | n/a | n/a | 950 | n/a | n/a | n/a | n/a |
Georgetown University; Duke University US Patent | Assay Description The functional properties of the ligands were determined by 86Rb+ efflux assays in cells expressing α3β4 and α4β2 nAChR subtypes.... | US Patent US9303017 (2016) BindingDB Entry DOI: 10.7270/Q25X27SF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Neuronal acetylcholine receptor Alpha-3/Beta-4 (Rattus norvegicus (Rat)) | BDBM221048 (US9284322, varenicline | US9303017, Varenicline) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid PDB UniChem | US Patent | n/a | n/a | n/a | n/a | 2.20E+4 | n/a | n/a | n/a | n/a |
Georgetown University; Duke University US Patent | Assay Description The functional properties of the ligands were determined by 86Rb+ efflux assays in cells expressing α3β4 and α4β2 nAChR subtypes.... | US Patent US9303017 (2016) BindingDB Entry DOI: 10.7270/Q25X27SF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||