 Found 14 hits for monomerid = 2337
Found 14 hits for monomerid = 2337 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
HIV-1 Reverse Transcriptase
(Human immunodeficiency virus type 1) | BDBM2337
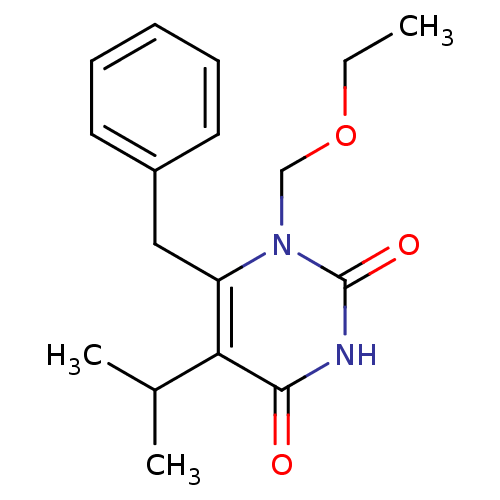
(6-benzyl-1-(ethoxymethyl)-5-(propan-2-yl)-1,2,3,4-...)Show InChI InChI=1S/C17H22N2O3/c1-4-22-11-19-14(10-13-8-6-5-7-9-13)15(12(2)3)16(20)18-17(19)21/h5-9,12H,4,10-11H2,1-3H3,(H,18,20,21) | PDB
MMDB
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | 7.8 | 37 |
Universita degli Studi di Roma La Sapienza
| Assay Description
The IC50 of reverse transcriptase is the concentration that inhibits 50% of recombinant HIV-1 RT RNA-directed DNA polymerase activity in vitro. |
J Med Chem 44: 2544-54 (2001)
Article DOI: 10.1021/jm010853h
BindingDB Entry DOI: 10.7270/Q2CN724V |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
HIV-1 Reverse Transcriptase
(Human immunodeficiency virus type 1) | BDBM2337

(6-benzyl-1-(ethoxymethyl)-5-(propan-2-yl)-1,2,3,4-...)Show InChI InChI=1S/C17H22N2O3/c1-4-22-11-19-14(10-13-8-6-5-7-9-13)15(12(2)3)16(20)18-17(19)21/h5-9,12H,4,10-11H2,1-3H3,(H,18,20,21) | PDB
MMDB
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Universita degli Studi di Roma La Sapienza
| Assay Description
The IC50 of reverse transcriptase is the concentration that inhibits 50% of recombinant HIV-1 RT RNA-directed DNA polymerase activity in vitro. |
J Med Chem 42: 619-27 (1999)
Article DOI: 10.1021/jm980260f
BindingDB Entry DOI: 10.7270/Q2445JPQ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
HIV-1 Reverse Transcriptase
(Human immunodeficiency virus type 1) | BDBM2337

(6-benzyl-1-(ethoxymethyl)-5-(propan-2-yl)-1,2,3,4-...)Show InChI InChI=1S/C17H22N2O3/c1-4-22-11-19-14(10-13-8-6-5-7-9-13)15(12(2)3)16(20)18-17(19)21/h5-9,12H,4,10-11H2,1-3H3,(H,18,20,21) | PDB
MMDB
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | 7.8 | 37 |
Universita degli Studi di Roma La Sapienza
| Assay Description
The IC50 of reverse transcriptase is the concentration that inhibits 50% of recombinant HIV-1 RT RNA-directed DNA polymerase activity in vitro. |
J Med Chem 47: 928-34 (2004)
Article DOI: 10.1021/jm0309856
BindingDB Entry DOI: 10.7270/Q26W988R |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Human immunodeficiency virus type 1 reverse transcriptase
(Human immunodeficiency virus 1) | BDBM2337

(6-benzyl-1-(ethoxymethyl)-5-(propan-2-yl)-1,2,3,4-...)Show InChI InChI=1S/C17H22N2O3/c1-4-22-11-19-14(10-13-8-6-5-7-9-13)15(12(2)3)16(20)18-17(19)21/h5-9,12H,4,10-11H2,1-3H3,(H,18,20,21) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
PubMed
| n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Inhibitory activity against recombinant HIV-1 reverse transcriptase (rRT) |
Bioorg Med Chem Lett 9: 2721-6 (1999)
BindingDB Entry DOI: 10.7270/Q2D50M49 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Human immunodeficiency virus type 1 reverse transcriptase
(Human immunodeficiency virus 1) | BDBM2337

(6-benzyl-1-(ethoxymethyl)-5-(propan-2-yl)-1,2,3,4-...)Show InChI InChI=1S/C17H22N2O3/c1-4-22-11-19-14(10-13-8-6-5-7-9-13)15(12(2)3)16(20)18-17(19)21/h5-9,12H,4,10-11H2,1-3H3,(H,18,20,21) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
PubMed
| n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Inhibition of recombinant reverse transcriptase (RT) in cell-free Quan-T-RT assay system |
Bioorg Med Chem Lett 9: 3411-6 (2000)
BindingDB Entry DOI: 10.7270/Q2SX6CD8 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Human immunodeficiency virus type 1 reverse transcriptase
(Human immunodeficiency virus 1) | BDBM2337

(6-benzyl-1-(ethoxymethyl)-5-(propan-2-yl)-1,2,3,4-...)Show InChI InChI=1S/C17H22N2O3/c1-4-22-11-19-14(10-13-8-6-5-7-9-13)15(12(2)3)16(20)18-17(19)21/h5-9,12H,4,10-11H2,1-3H3,(H,18,20,21) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
PubMed
| n/a | n/a | n/a | n/a | 20 | n/a | n/a | n/a | n/a |
Yale University
Curated by ChEMBL
| Assay Description
Effective concentration required against L100I mutant HIV-1 reverse transcriptase |
Bioorg Med Chem Lett 11: 2799-802 (2001)
BindingDB Entry DOI: 10.7270/Q2J103P4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Reverse transcriptase
(Human immunodeficiency virus 1) | BDBM2337

(6-benzyl-1-(ethoxymethyl)-5-(propan-2-yl)-1,2,3,4-...)Show InChI InChI=1S/C17H22N2O3/c1-4-22-11-19-14(10-13-8-6-5-7-9-13)15(12(2)3)16(20)18-17(19)21/h5-9,12H,4,10-11H2,1-3H3,(H,18,20,21) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
Peking University
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 reverse transcriptase using poly(ra)/oligo(dT)15 homopolymer template as substrate after 1 hr |
J Med Chem 55: 2242-50 (2012)
Article DOI: 10.1021/jm201506e
BindingDB Entry DOI: 10.7270/Q22N554J |
More data for this
Ligand-Target Pair | |
Human immunodeficiency virus type 1 reverse transcriptase
(Human immunodeficiency virus 1) | BDBM2337

(6-benzyl-1-(ethoxymethyl)-5-(propan-2-yl)-1,2,3,4-...)Show InChI InChI=1S/C17H22N2O3/c1-4-22-11-19-14(10-13-8-6-5-7-9-13)15(12(2)3)16(20)18-17(19)21/h5-9,12H,4,10-11H2,1-3H3,(H,18,20,21) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 4.20 | n/a | n/a | n/a | n/a |
Université d'Orléans
Curated by ChEMBL
| Assay Description
Inhibitory concentration against HIV-1 reverse transcriptase |
J Med Chem 40: 4257-64 (1998)
Article DOI: 10.1021/jm970110p
BindingDB Entry DOI: 10.7270/Q2WS8SC3 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Human immunodeficiency virus type 1 reverse transcriptase
(Human immunodeficiency virus 1) | BDBM2337

(6-benzyl-1-(ethoxymethyl)-5-(propan-2-yl)-1,2,3,4-...)Show InChI InChI=1S/C17H22N2O3/c1-4-22-11-19-14(10-13-8-6-5-7-9-13)15(12(2)3)16(20)18-17(19)21/h5-9,12H,4,10-11H2,1-3H3,(H,18,20,21) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
PubMed
| n/a | n/a | n/a | n/a | 4 | n/a | n/a | n/a | n/a |
Yale University
Curated by ChEMBL
| Assay Description
Inhibition of HIV-1 reverse transcriptase at 37 degree centigrade |
J Med Chem 44: 145-54 (2001)
BindingDB Entry DOI: 10.7270/Q2VX0FRX |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Human immunodeficiency virus type 1 reverse transcriptase
(Human immunodeficiency virus 1) | BDBM2337

(6-benzyl-1-(ethoxymethyl)-5-(propan-2-yl)-1,2,3,4-...)Show InChI InChI=1S/C17H22N2O3/c1-4-22-11-19-14(10-13-8-6-5-7-9-13)15(12(2)3)16(20)18-17(19)21/h5-9,12H,4,10-11H2,1-3H3,(H,18,20,21) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
PubMed
| n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Inhibition of purified recombinant HIV-1 reverse transcriptase |
Bioorg Med Chem Lett 9: 1593-8 (1999)
BindingDB Entry DOI: 10.7270/Q27H1K3Q |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Human immunodeficiency virus type 1 reverse transcriptase
(Human immunodeficiency virus 1) | BDBM2337

(6-benzyl-1-(ethoxymethyl)-5-(propan-2-yl)-1,2,3,4-...)Show InChI InChI=1S/C17H22N2O3/c1-4-22-11-19-14(10-13-8-6-5-7-9-13)15(12(2)3)16(20)18-17(19)21/h5-9,12H,4,10-11H2,1-3H3,(H,18,20,21) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Universita degli Studi di Roma La Sapienza
| Assay Description
Inhibitory concentration required to inhibit the HIV-1 reverse transcriptase activity |
J Med Chem 47: 928-34 (2004)
Article DOI: 10.1021/jm0309856
BindingDB Entry DOI: 10.7270/Q26W988R |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Human immunodeficiency virus type 1 reverse transcriptase
(Human immunodeficiency virus 1) | BDBM2337

(6-benzyl-1-(ethoxymethyl)-5-(propan-2-yl)-1,2,3,4-...)Show InChI InChI=1S/C17H22N2O3/c1-4-22-11-19-14(10-13-8-6-5-7-9-13)15(12(2)3)16(20)18-17(19)21/h5-9,12H,4,10-11H2,1-3H3,(H,18,20,21) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
PubMed
| n/a | n/a | n/a | n/a | 2 | n/a | n/a | n/a | n/a |
Yale University
Curated by ChEMBL
| Assay Description
Effective concentration required against wild type HIV-1 reverse transcriptase |
Bioorg Med Chem Lett 11: 2799-802 (2001)
BindingDB Entry DOI: 10.7270/Q2J103P4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Reverse transcriptase
(Human immunodeficiency virus 1) | BDBM2337

(6-benzyl-1-(ethoxymethyl)-5-(propan-2-yl)-1,2,3,4-...)Show InChI InChI=1S/C17H22N2O3/c1-4-22-11-19-14(10-13-8-6-5-7-9-13)15(12(2)3)16(20)18-17(19)21/h5-9,12H,4,10-11H2,1-3H3,(H,18,20,21) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
Peking University
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 reverse transcriptase by ELISA |
Bioorg Med Chem 18: 3231-7 (2010)
Article DOI: 10.1016/j.bmc.2010.03.025
BindingDB Entry DOI: 10.7270/Q2057JQ1 |
More data for this
Ligand-Target Pair | |
Human immunodeficiency virus type 1 reverse transcriptase
(Human immunodeficiency virus 1) | BDBM2337

(6-benzyl-1-(ethoxymethyl)-5-(propan-2-yl)-1,2,3,4-...)Show InChI InChI=1S/C17H22N2O3/c1-4-22-11-19-14(10-13-8-6-5-7-9-13)15(12(2)3)16(20)18-17(19)21/h5-9,12H,4,10-11H2,1-3H3,(H,18,20,21) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Oxford
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against HIV-1 reverse transcriptase |
J Med Chem 39: 1589-600 (1996)
Article DOI: 10.1021/jm960056x
BindingDB Entry DOI: 10.7270/Q23X85QC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
