

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM23916 3-amidinophenylalanine deriv., 12::3-amino-N-(3-{[(2S)-1-[4-(2-carbamimidamidoethyl)piperidin-1-yl]-3-(3-carbamimidoylphenyl)-1-oxopropan-2-yl]sulfamoyl}phenyl)propanamide::CHEMBL378657
SMILES: [#7]-[#6]-[#6]-[#6](=O)-[#7]-c1cccc(c1)S(=O)(=O)[#7]-[#6@@H](-[#6]-c1cccc(c1)-[#6](-[#7])=[#7])-[#6](=O)-[#7]-1-[#6]-[#6]-[#6](-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6]-[#6]-1
InChI Key: InChIKey=ZEWBVTHVYJHWAH-QHCPKHFHSA-N
Data: 10 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hepatocyte growth factor activator/Serine protease hepsin/Suppressor of tumorigenicity 14 protein (Homo sapiens (Human)) | BDBM23916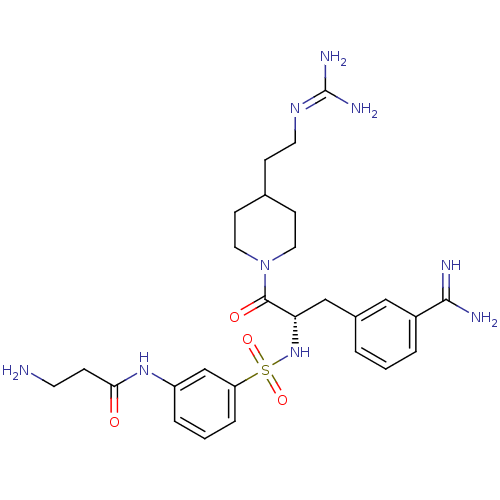 (3-amidinophenylalanine deriv., 12 | 3-amino-N-(3-{...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Curacyte Chemistry GmbH | Assay Description The measurements were carried out on a microplate reader. Two concentrations of the substrate and five concentrations of the inhibitor were used. Af... | J Med Chem 49: 4116-26 (2006) Article DOI: 10.1021/jm051272l BindingDB Entry DOI: 10.7270/Q21C1V64 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor activator/Serine protease hepsin/Suppressor of tumorigenicity 14 protein (Homo sapiens (Human)) | BDBM23916 (3-amidinophenylalanine deriv., 12 | 3-amino-N-(3-{...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Medicines Company (Leipzig) GmbH Curated by ChEMBL | Assay Description Inhibition of matriptase (unknown origin) | Bioorg Med Chem Lett 19: 1960-5 (2009) Article DOI: 10.1016/j.bmcl.2009.02.047 BindingDB Entry DOI: 10.7270/Q2J96681 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Urokinase-type plasminogen activator (Homo sapiens (Human)) | BDBM23916 (3-amidinophenylalanine deriv., 12 | 3-amino-N-(3-{...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Curacyte Chemistry GmbH | Assay Description The measurements were carried out on a microplate reader. Two concentrations of the substrate and five concentrations of the inhibitor were used. Af... | J Med Chem 49: 4116-26 (2006) Article DOI: 10.1021/jm051272l BindingDB Entry DOI: 10.7270/Q21C1V64 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Urokinase-type plasminogen activator (Homo sapiens (Human)) | BDBM23916 (3-amidinophenylalanine deriv., 12 | 3-amino-N-(3-{...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Medicines Company (Leipzig) GmbH Curated by ChEMBL | Assay Description Inhibition of uPA (unknown origin) | Bioorg Med Chem Lett 19: 1960-5 (2009) Article DOI: 10.1016/j.bmcl.2009.02.047 BindingDB Entry DOI: 10.7270/Q2J96681 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasminogen (Homo sapiens (Human)) | BDBM23916 (3-amidinophenylalanine deriv., 12 | 3-amino-N-(3-{...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Curacyte Chemistry GmbH | Assay Description The measurements were carried out on a microplate reader. Two concentrations of the substrate and five concentrations of the inhibitor were used. Af... | J Med Chem 49: 4116-26 (2006) Article DOI: 10.1021/jm051272l BindingDB Entry DOI: 10.7270/Q21C1V64 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasminogen (Homo sapiens (Human)) | BDBM23916 (3-amidinophenylalanine deriv., 12 | 3-amino-N-(3-{...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Medicines Company (Leipzig) GmbH Curated by ChEMBL | Assay Description Inhibition of plasmin (unknown origin) | Bioorg Med Chem Lett 19: 1960-5 (2009) Article DOI: 10.1016/j.bmcl.2009.02.047 BindingDB Entry DOI: 10.7270/Q2J96681 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Thrombin (Bos taurus (Bovine)) | BDBM23916 (3-amidinophenylalanine deriv., 12 | 3-amino-N-(3-{...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 6.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Curacyte Chemistry GmbH | Assay Description The measurements were carried out on a microplate reader. Two concentrations of the substrate and five concentrations of the inhibitor were used. Af... | J Med Chem 49: 4116-26 (2006) Article DOI: 10.1021/jm051272l BindingDB Entry DOI: 10.7270/Q21C1V64 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM23916 (3-amidinophenylalanine deriv., 12 | 3-amino-N-(3-{...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 6.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Medicines Company (Leipzig) GmbH Curated by ChEMBL | Assay Description Inhibition of thrombin (unknown origin) | Bioorg Med Chem Lett 19: 1960-5 (2009) Article DOI: 10.1016/j.bmcl.2009.02.047 BindingDB Entry DOI: 10.7270/Q2J96681 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM23916 (3-amidinophenylalanine deriv., 12 | 3-amino-N-(3-{...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 9.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Medicines Company (Leipzig) GmbH Curated by ChEMBL | Assay Description Inhibition of factor 10a (unknown origin) | Bioorg Med Chem Lett 19: 1960-5 (2009) Article DOI: 10.1016/j.bmcl.2009.02.047 BindingDB Entry DOI: 10.7270/Q2J96681 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Factor Xa (fXa) (Bos taurus) | BDBM23916 (3-amidinophenylalanine deriv., 12 | 3-amino-N-(3-{...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 9.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Curacyte Chemistry GmbH | Assay Description The measurements were carried out on a microplate reader. Two concentrations of the substrate and five concentrations of the inhibitor were used. Af... | J Med Chem 49: 4116-26 (2006) Article DOI: 10.1021/jm051272l BindingDB Entry DOI: 10.7270/Q21C1V64 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||