

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM2580 K252a::methyl (15R,16S,18S)-16-hydroxy-15-methyl-3-oxo-28-oxa-4,14,19-triazaoctacyclo[12.11.2.1^{15,18}.0^{2,6}.0^{7,27}.0^{8,13}.0^{19,26}.0^{20,25}]octacosa-1(26),2(6),7(27),8(13),9,11,20(25),21,23-nonaene-16-carboxylate
SMILES: COC(=O)[C@]1(O)C[C@@H]2O[C@@]1(C)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13
InChI Key: InChIKey=KOZFSFOOLUUIGY-CYBHFKQVSA-N
PDB links: 1 PDB ID contains this monomer as substructures. 1 PDB ID contains inhibitors having a similarity of 90% to this monomer.
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Calcium/calmodulin-dependent protein kinase kinase 2 (Homo sapiens (Human)) | BDBM2580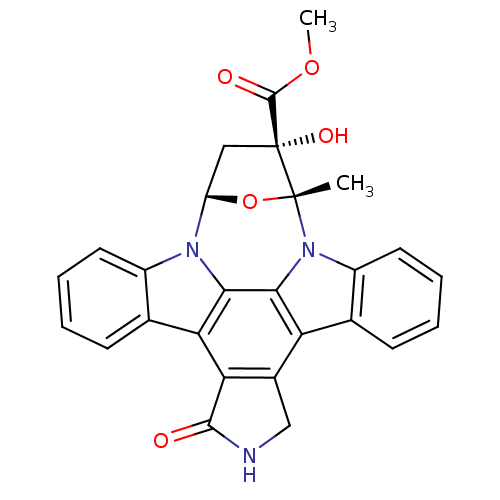 (K252a | methyl (15R,16S,18S)-16-hydroxy-15-methyl-...) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Oxford Curated by ChEMBL | Assay Description Inhibition of CaM-KKbeta | Proc Natl Acad Sci USA 104: 20523-8 (2007) Checked by Author Article DOI: 10.1073/pnas.0708800104 BindingDB Entry DOI: 10.7270/Q2DB82RH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Myosin light chain kinase, smooth muscle (Homo sapiens (Human)) | BDBM2580 (K252a | methyl (15R,16S,18S)-16-hydroxy-15-methyl-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | 17 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Oxford Curated by ChEMBL | Assay Description Inhibition of human myosin light chain kinase | Proc Natl Acad Sci USA 104: 20523-8 (2007) Checked by Author Article DOI: 10.1073/pnas.0708800104 BindingDB Entry DOI: 10.7270/Q2DB82RH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin receptor (Homo sapiens (Human)) | BDBM2580 (K252a | methyl (15R,16S,18S)-16-hydroxy-15-methyl-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 210 | n/a | n/a | n/a | n/a | 7.4 | 22 |
Merck Research Laboratories | Assay Description The assay uses purified enzyme interacting with biotinylated peptide substrate.HTRF is based on the proximity of europium cryptate (donor fluorophore... | Biochemistry 44: 9430-40 (2005) Article DOI: 10.1021/bi0500628 BindingDB Entry DOI: 10.7270/Q2BV7DV5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protein Kinase C (Rattus norvegicus (rat)) | BDBM2580 (K252a | methyl (15R,16S,18S)-16-hydroxy-15-methyl-...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 470 | n/a | n/a | n/a | n/a | 7.5 | 30 |
Roche Products Limited | Assay Description The activity of PKC, activated by phosphatidylerine and Ca2+, is measured by its ability to transfer phosphate from [gamma-32P]ATP to lysine-rich his... | J Med Chem 35: 177-84 (1992) Article DOI: 10.1021/jm00079a024 BindingDB Entry DOI: 10.7270/Q2K64G8V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin-like growth factor I receptor (Homo sapiens (Human)) | BDBM2580 (K252a | methyl (15R,16S,18S)-16-hydroxy-15-methyl-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 470 | n/a | n/a | n/a | n/a | 7.4 | 22 |
Merck Research Laboratories | Assay Description The assay uses purified enzyme interacting with biotinylated peptide substrate.HTRF is based on the proximity of europium cryptate (donor fluorophore... | Biochemistry 44: 9430-40 (2005) Article DOI: 10.1021/bi0500628 BindingDB Entry DOI: 10.7270/Q2BV7DV5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||