

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM25895 N-heterocyclic dipeptide aldehyde, 13::methyl 5-{[(1S)-2-methyl-1-{[(2S)-4-methyl-1-oxopentan-2-yl]carbamoyl}propyl]carbamoyl}-1H-pyrrole-2-carboxylate
SMILES: COC(=O)c1ccc([nH]1)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(C)C)C=O
InChI Key: InChIKey=OHADBCSEOORETK-WFASDCNBSA-N
Data: 2 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Calpain-2 (Ovis aries (sheep)) | BDBM25895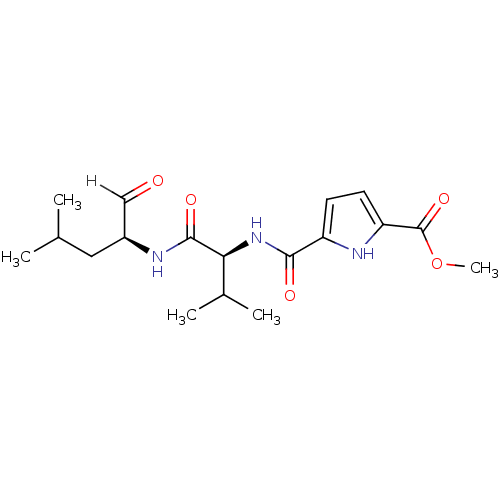 (N-heterocyclic dipeptide aldehyde, 13 | methyl 5-{...) | PDB MMDB KEGG UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 140 | n/a | n/a | n/a | n/a | 7.5 | 25 |
University of Canterbury | Assay Description Calpain inhibition assays were performed in 96-well black plates. The reaction was initiated by the addition of substrate solution. The progress of t... | Bioorg Med Chem 16: 6911-23 (2008) Article DOI: 10.1016/j.bmc.2008.05.048 BindingDB Entry DOI: 10.7270/Q2C53J5R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Calpain-1 (Ovis aries (sheep)) | BDBM25895 (N-heterocyclic dipeptide aldehyde, 13 | methyl 5-{...) | PDB MMDB KEGG UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 290 | n/a | n/a | n/a | n/a | 7.5 | 25 |
University of Canterbury | Assay Description Calpain inhibition assays were performed in 96-well black plates. The reaction was initiated by the addition of substrate solution. The progress of t... | Bioorg Med Chem 16: 6911-23 (2008) Article DOI: 10.1016/j.bmc.2008.05.048 BindingDB Entry DOI: 10.7270/Q2C53J5R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||