 Found 13 hits for monomerid = 26105
Found 13 hits for monomerid = 26105 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM26105
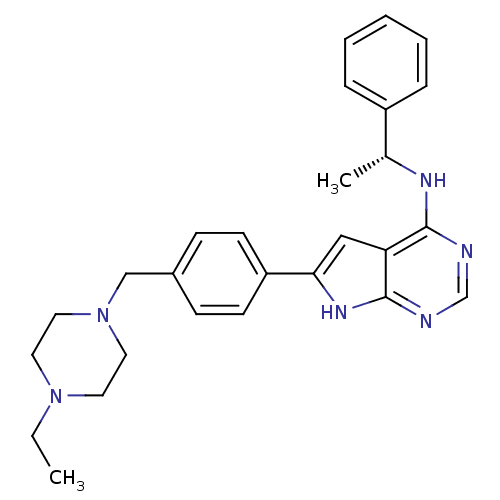
(6-{4-[(4-ethylpiperazin-1-yl)methyl]phenyl}-N-[(1R...)Show SMILES CCN1CCN(Cc2ccc(cc2)-c2cc3c(N[C@H](C)c4ccccc4)ncnc3[nH]2)CC1 |r| Show InChI InChI=1S/C27H32N6/c1-3-32-13-15-33(16-14-32)18-21-9-11-23(12-10-21)25-17-24-26(28-19-29-27(24)31-25)30-20(2)22-7-5-4-6-8-22/h4-12,17,19-20H,3,13-16,18H2,1-2H3,(H2,28,29,30,31)/t20-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | 10.9 | n/a | n/a | n/a | 7.4 | 22 |
Harvard Medical School
| Assay Description
The fluorescence buffer was degassed and aerated with pure nitrogen gas to remove dissolved oxygen. The assay was carried out on a FluoroMax-2 fluoro... |
Cancer Cell 11: 217-27 (2007)
Article DOI: 10.1016/j.ccr.2006.12.017
BindingDB Entry DOI: 10.7270/Q2JQ0ZBW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
EGF-R Tyrosine Kinase Mutant (G719S)
(Homo sapiens (Human)) | BDBM26105

(6-{4-[(4-ethylpiperazin-1-yl)methyl]phenyl}-N-[(1R...)Show SMILES CCN1CCN(Cc2ccc(cc2)-c2cc3c(N[C@H](C)c4ccccc4)ncnc3[nH]2)CC1 |r| Show InChI InChI=1S/C27H32N6/c1-3-32-13-15-33(16-14-32)18-21-9-11-23(12-10-21)25-17-24-26(28-19-29-27(24)31-25)30-20(2)22-7-5-4-6-8-22/h4-12,17,19-20H,3,13-16,18H2,1-2H3,(H2,28,29,30,31)/t20-/m1/s1 | PDB
MMDB
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | 11.3 | n/a | n/a | n/a | 7.4 | 22 |
Harvard Medical School
| Assay Description
The fluorescence buffer was degassed and aerated with pure nitrogen gas to remove dissolved oxygen. The assay was carried out on a FluoroMax-2 fluoro... |
Cancer Cell 11: 217-27 (2007)
Article DOI: 10.1016/j.ccr.2006.12.017
BindingDB Entry DOI: 10.7270/Q2JQ0ZBW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
EGF-R Tyrosine Kinase Mutant (L858R)
(Homo sapiens (Human)) | BDBM26105

(6-{4-[(4-ethylpiperazin-1-yl)methyl]phenyl}-N-[(1R...)Show SMILES CCN1CCN(Cc2ccc(cc2)-c2cc3c(N[C@H](C)c4ccccc4)ncnc3[nH]2)CC1 |r| Show InChI InChI=1S/C27H32N6/c1-3-32-13-15-33(16-14-32)18-21-9-11-23(12-10-21)25-17-24-26(28-19-29-27(24)31-25)30-20(2)22-7-5-4-6-8-22/h4-12,17,19-20H,3,13-16,18H2,1-2H3,(H2,28,29,30,31)/t20-/m1/s1 | PDB
MMDB
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | 1.70 | n/a | n/a | n/a | 7.4 | 22 |
Harvard Medical School
| Assay Description
The fluorescence buffer was degassed and aerated with pure nitrogen gas to remove dissolved oxygen. The assay was carried out on a FluoroMax-2 fluoro... |
Cancer Cell 11: 217-27 (2007)
Article DOI: 10.1016/j.ccr.2006.12.017
BindingDB Entry DOI: 10.7270/Q2JQ0ZBW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM26105

(6-{4-[(4-ethylpiperazin-1-yl)methyl]phenyl}-N-[(1R...)Show SMILES CCN1CCN(Cc2ccc(cc2)-c2cc3c(N[C@H](C)c4ccccc4)ncnc3[nH]2)CC1 |r| Show InChI InChI=1S/C27H32N6/c1-3-32-13-15-33(16-14-32)18-21-9-11-23(12-10-21)25-17-24-26(28-19-29-27(24)31-25)30-20(2)22-7-5-4-6-8-22/h4-12,17,19-20H,3,13-16,18H2,1-2H3,(H2,28,29,30,31)/t20-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | 5.30 | n/a | n/a | n/a | 7.4 | 22 |
Harvard Medical School
| Assay Description
The fluorescence buffer was degassed and aerated with pure nitrogen gas to remove dissolved oxygen. The assay was carried out on a FluoroMax-2 fluoro... |
Proc Natl Acad Sci U S A 105: 2070-5 (2008)
Article DOI: 10.1073/pnas.0709662105
BindingDB Entry DOI: 10.7270/Q2DZ06MJ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
EGF-R Tyrosine Kinase Mutant (T790M)
(Homo sapiens (Human)) | BDBM26105

(6-{4-[(4-ethylpiperazin-1-yl)methyl]phenyl}-N-[(1R...)Show SMILES CCN1CCN(Cc2ccc(cc2)-c2cc3c(N[C@H](C)c4ccccc4)ncnc3[nH]2)CC1 |r| Show InChI InChI=1S/C27H32N6/c1-3-32-13-15-33(16-14-32)18-21-9-11-23(12-10-21)25-17-24-26(28-19-29-27(24)31-25)30-20(2)22-7-5-4-6-8-22/h4-12,17,19-20H,3,13-16,18H2,1-2H3,(H2,28,29,30,31)/t20-/m1/s1 | PDB
MMDB
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | 27.6 | n/a | n/a | n/a | 7.4 | 22 |
Harvard Medical School
| Assay Description
The fluorescence buffer was degassed and aerated with pure nitrogen gas to remove dissolved oxygen. The assay was carried out on a FluoroMax-2 fluoro... |
Proc Natl Acad Sci U S A 105: 2070-5 (2008)
Article DOI: 10.1073/pnas.0709662105
BindingDB Entry DOI: 10.7270/Q2DZ06MJ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
EGF-R Tyrosine Kinase Mutant (L858R)
(Homo sapiens (Human)) | BDBM26105

(6-{4-[(4-ethylpiperazin-1-yl)methyl]phenyl}-N-[(1R...)Show SMILES CCN1CCN(Cc2ccc(cc2)-c2cc3c(N[C@H](C)c4ccccc4)ncnc3[nH]2)CC1 |r| Show InChI InChI=1S/C27H32N6/c1-3-32-13-15-33(16-14-32)18-21-9-11-23(12-10-21)25-17-24-26(28-19-29-27(24)31-25)30-20(2)22-7-5-4-6-8-22/h4-12,17,19-20H,3,13-16,18H2,1-2H3,(H2,28,29,30,31)/t20-/m1/s1 | PDB
MMDB
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | 1.10 | n/a | n/a | n/a | 7.4 | 22 |
Harvard Medical School
| Assay Description
The fluorescence buffer was degassed and aerated with pure nitrogen gas to remove dissolved oxygen. The assay was carried out on a FluoroMax-2 fluoro... |
Proc Natl Acad Sci U S A 105: 2070-5 (2008)
Article DOI: 10.1073/pnas.0709662105
BindingDB Entry DOI: 10.7270/Q2DZ06MJ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein kinase Yes
(Homo sapiens (Human)) | BDBM26105

(6-{4-[(4-ethylpiperazin-1-yl)methyl]phenyl}-N-[(1R...)Show SMILES CCN1CCN(Cc2ccc(cc2)-c2cc3c(N[C@H](C)c4ccccc4)ncnc3[nH]2)CC1 |r| Show InChI InChI=1S/C27H32N6/c1-3-32-13-15-33(16-14-32)18-21-9-11-23(12-10-21)25-17-24-26(28-19-29-27(24)31-25)30-20(2)22-7-5-4-6-8-22/h4-12,17,19-20H,3,13-16,18H2,1-2H3,(H2,28,29,30,31)/t20-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Frederick National Laboratory for Cancer Research
Curated by ChEMBL
| Assay Description
Inhibition of Yes1 (unknown origin) assessed as kinase-dependent enzymatic production of ADP from ATP using coupled luminescence-based reaction by AD... |
Bioorg Med Chem Lett 23: 4398-403 (2013)
Article DOI: 10.1016/j.bmcl.2013.05.072
BindingDB Entry DOI: 10.7270/Q2BR8W3F |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret
(Homo sapiens (Human)) | BDBM26105

(6-{4-[(4-ethylpiperazin-1-yl)methyl]phenyl}-N-[(1R...)Show SMILES CCN1CCN(Cc2ccc(cc2)-c2cc3c(N[C@H](C)c4ccccc4)ncnc3[nH]2)CC1 |r| Show InChI InChI=1S/C27H32N6/c1-3-32-13-15-33(16-14-32)18-21-9-11-23(12-10-21)25-17-24-26(28-19-29-27(24)31-25)30-20(2)22-7-5-4-6-8-22/h4-12,17,19-20H,3,13-16,18H2,1-2H3,(H2,28,29,30,31)/t20-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 740 | n/a | n/a | n/a | n/a | n/a | n/a |
National Tsing Hua University
Curated by ChEMBL
| Assay Description
Inhibition of RET kinase |
Bioorg Med Chem Lett 21: 4490-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.003
BindingDB Entry DOI: 10.7270/Q2BV7H06 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM26105

(6-{4-[(4-ethylpiperazin-1-yl)methyl]phenyl}-N-[(1R...)Show SMILES CCN1CCN(Cc2ccc(cc2)-c2cc3c(N[C@H](C)c4ccccc4)ncnc3[nH]2)CC1 |r| Show InChI InChI=1S/C27H32N6/c1-3-32-13-15-33(16-14-32)18-21-9-11-23(12-10-21)25-17-24-26(28-19-29-27(24)31-25)30-20(2)22-7-5-4-6-8-22/h4-12,17,19-20H,3,13-16,18H2,1-2H3,(H2,28,29,30,31)/t20-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Norwegian University of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of EGFR by Z-LYTE assay |
Eur J Med Chem 46: 6002-14 (2011)
Article DOI: 10.1016/j.ejmech.2011.10.012
BindingDB Entry DOI: 10.7270/Q26M377V |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Vascular endothelial growth factor receptor 1
(Homo sapiens (Human)) | BDBM26105

(6-{4-[(4-ethylpiperazin-1-yl)methyl]phenyl}-N-[(1R...)Show SMILES CCN1CCN(Cc2ccc(cc2)-c2cc3c(N[C@H](C)c4ccccc4)ncnc3[nH]2)CC1 |r| Show InChI InChI=1S/C27H32N6/c1-3-32-13-15-33(16-14-32)18-21-9-11-23(12-10-21)25-17-24-26(28-19-29-27(24)31-25)30-20(2)22-7-5-4-6-8-22/h4-12,17,19-20H,3,13-16,18H2,1-2H3,(H2,28,29,30,31)/t20-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 59 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Genoa
Curated by ChEMBL
| Assay Description
Inhibition of VEGFR1 |
J Med Chem 55: 10797-822 (2012)
Article DOI: 10.1021/jm301085w
BindingDB Entry DOI: 10.7270/Q2BP03XZ |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM26105

(6-{4-[(4-ethylpiperazin-1-yl)methyl]phenyl}-N-[(1R...)Show SMILES CCN1CCN(Cc2ccc(cc2)-c2cc3c(N[C@H](C)c4ccccc4)ncnc3[nH]2)CC1 |r| Show InChI InChI=1S/C27H32N6/c1-3-32-13-15-33(16-14-32)18-21-9-11-23(12-10-21)25-17-24-26(28-19-29-27(24)31-25)30-20(2)22-7-5-4-6-8-22/h4-12,17,19-20H,3,13-16,18H2,1-2H3,(H2,28,29,30,31)/t20-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 77 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Genoa
Curated by ChEMBL
| Assay Description
Inhibition of VEGFR2 |
J Med Chem 55: 10797-822 (2012)
Article DOI: 10.1021/jm301085w
BindingDB Entry DOI: 10.7270/Q2BP03XZ |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Yes
(Homo sapiens (Human)) | BDBM26105

(6-{4-[(4-ethylpiperazin-1-yl)methyl]phenyl}-N-[(1R...)Show SMILES CCN1CCN(Cc2ccc(cc2)-c2cc3c(N[C@H](C)c4ccccc4)ncnc3[nH]2)CC1 |r| Show InChI InChI=1S/C27H32N6/c1-3-32-13-15-33(16-14-32)18-21-9-11-23(12-10-21)25-17-24-26(28-19-29-27(24)31-25)30-20(2)22-7-5-4-6-8-22/h4-12,17,19-20H,3,13-16,18H2,1-2H3,(H2,28,29,30,31)/t20-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Frederick National Laboratory for Cancer Research
Curated by ChEMBL
| Assay Description
Inhibition of Yes1 (unknown origin) by [gamma-33P]-ATP radiolabeled enzyme activity assay |
Bioorg Med Chem Lett 23: 4398-403 (2013)
Article DOI: 10.1016/j.bmcl.2013.05.072
BindingDB Entry DOI: 10.7270/Q2BR8W3F |
More data for this
Ligand-Target Pair | |
EGF-R Tyrosine Kinase Mutant (L858R/T790M)
(Homo sapiens (Human)) | BDBM26105

(6-{4-[(4-ethylpiperazin-1-yl)methyl]phenyl}-N-[(1R...)Show SMILES CCN1CCN(Cc2ccc(cc2)-c2cc3c(N[C@H](C)c4ccccc4)ncnc3[nH]2)CC1 |r| Show InChI InChI=1S/C27H32N6/c1-3-32-13-15-33(16-14-32)18-21-9-11-23(12-10-21)25-17-24-26(28-19-29-27(24)31-25)30-20(2)22-7-5-4-6-8-22/h4-12,17,19-20H,3,13-16,18H2,1-2H3,(H2,28,29,30,31)/t20-/m1/s1 | PDB
MMDB
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | 18.6 | n/a | n/a | n/a | 7.4 | 22 |
Harvard Medical School
| Assay Description
The fluorescence buffer was degassed and aerated with pure nitrogen gas to remove dissolved oxygen. The assay was carried out on a FluoroMax-2 fluoro... |
Proc Natl Acad Sci U S A 105: 2070-5 (2008)
Article DOI: 10.1073/pnas.0709662105
BindingDB Entry DOI: 10.7270/Q2DZ06MJ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data