 Found 65 hits for monomerid = 26948
Found 65 hits for monomerid = 26948 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Thromboxane A2 receptor
(Homo sapiens (Human)) | BDBM26948
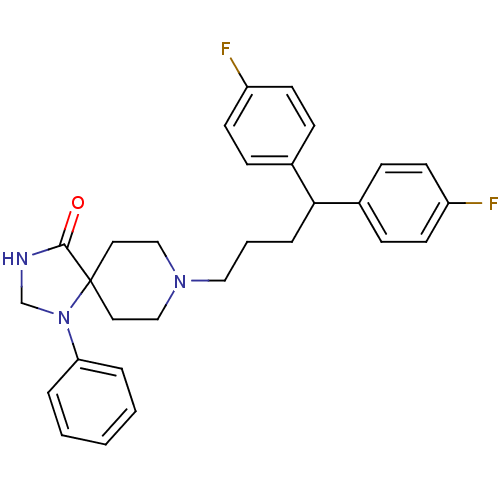
(8-[4,4-bis(4-fluorophenyl)butyl]-1-phenyl-1,3,8-tr...)Show SMILES Fc1ccc(cc1)C(CCCN1CCC2(CC1)N(CNC2=O)c1ccccc1)c1ccc(F)cc1 Show InChI InChI=1S/C29H31F2N3O/c30-24-12-8-22(9-13-24)27(23-10-14-25(31)15-11-23)7-4-18-33-19-16-29(17-20-33)28(35)32-21-34(29)26-5-2-1-3-6-26/h1-3,5-6,8-15,27H,4,7,16-21H2,(H,32,35) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
Psychopharmacology (Berl) 124: 57-73 (1996)
Article DOI: 10.1007/bf02245606
BindingDB Entry DOI: 10.7270/Q2610XV6 |
More data for this
Ligand-Target Pair | |
D(1A) dopamine receptor
(BOVINE) | BDBM26948

(8-[4,4-bis(4-fluorophenyl)butyl]-1-phenyl-1,3,8-tr...)Show SMILES Fc1ccc(cc1)C(CCCN1CCC2(CC1)N(CNC2=O)c1ccccc1)c1ccc(F)cc1 Show InChI InChI=1S/C29H31F2N3O/c30-24-12-8-22(9-13-24)27(23-10-14-25(31)15-11-23)7-4-18-33-19-16-29(17-20-33)28(35)32-21-34(29)26-5-2-1-3-6-26/h1-3,5-6,8-15,27H,4,7,16-21H2,(H,32,35) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by PDSP Ki Database
| |
J Neuropsychiatry Clin Neurosci 8: 223-6 (1996)
Article DOI: 10.1176/jnp.8.2.223
BindingDB Entry DOI: 10.7270/Q27S7M9V |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(BOVINE) | BDBM26948

(8-[4,4-bis(4-fluorophenyl)butyl]-1-phenyl-1,3,8-tr...)Show SMILES Fc1ccc(cc1)C(CCCN1CCC2(CC1)N(CNC2=O)c1ccccc1)c1ccc(F)cc1 Show InChI InChI=1S/C29H31F2N3O/c30-24-12-8-22(9-13-24)27(23-10-14-25(31)15-11-23)7-4-18-33-19-16-29(17-20-33)28(35)32-21-34(29)26-5-2-1-3-6-26/h1-3,5-6,8-15,27H,4,7,16-21H2,(H,32,35) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by PDSP Ki Database
| |
J Neuropsychiatry Clin Neurosci 8: 223-6 (1996)
Article DOI: 10.1176/jnp.8.2.223
BindingDB Entry DOI: 10.7270/Q27S7M9V |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM26948

(8-[4,4-bis(4-fluorophenyl)butyl]-1-phenyl-1,3,8-tr...)Show SMILES Fc1ccc(cc1)C(CCCN1CCC2(CC1)N(CNC2=O)c1ccccc1)c1ccc(F)cc1 Show InChI InChI=1S/C29H31F2N3O/c30-24-12-8-22(9-13-24)27(23-10-14-25(31)15-11-23)7-4-18-33-19-16-29(17-20-33)28(35)32-21-34(29)26-5-2-1-3-6-26/h1-3,5-6,8-15,27H,4,7,16-21H2,(H,32,35) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
ACADIA Pharmaceuticals
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 315: 1278-87 (2005)
Article DOI: 10.1124/jpet.105.092155
BindingDB Entry DOI: 10.7270/Q2Z60MNB |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM26948

(8-[4,4-bis(4-fluorophenyl)butyl]-1-phenyl-1,3,8-tr...)Show SMILES Fc1ccc(cc1)C(CCCN1CCC2(CC1)N(CNC2=O)c1ccccc1)c1ccc(F)cc1 Show InChI InChI=1S/C29H31F2N3O/c30-24-12-8-22(9-13-24)27(23-10-14-25(31)15-11-23)7-4-18-33-19-16-29(17-20-33)28(35)32-21-34(29)26-5-2-1-3-6-26/h1-3,5-6,8-15,27H,4,7,16-21H2,(H,32,35) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
ACADIA Pharmaceuticals
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 315: 1278-87 (2005)
Article DOI: 10.1124/jpet.105.092155
BindingDB Entry DOI: 10.7270/Q2Z60MNB |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM26948

(8-[4,4-bis(4-fluorophenyl)butyl]-1-phenyl-1,3,8-tr...)Show SMILES Fc1ccc(cc1)C(CCCN1CCC2(CC1)N(CNC2=O)c1ccccc1)c1ccc(F)cc1 Show InChI InChI=1S/C29H31F2N3O/c30-24-12-8-22(9-13-24)27(23-10-14-25(31)15-11-23)7-4-18-33-19-16-29(17-20-33)28(35)32-21-34(29)26-5-2-1-3-6-26/h1-3,5-6,8-15,27H,4,7,16-21H2,(H,32,35) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
ACADIA Pharmaceuticals
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 315: 1278-87 (2005)
Article DOI: 10.1124/jpet.105.092155
BindingDB Entry DOI: 10.7270/Q2Z60MNB |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM26948

(8-[4,4-bis(4-fluorophenyl)butyl]-1-phenyl-1,3,8-tr...)Show SMILES Fc1ccc(cc1)C(CCCN1CCC2(CC1)N(CNC2=O)c1ccccc1)c1ccc(F)cc1 Show InChI InChI=1S/C29H31F2N3O/c30-24-12-8-22(9-13-24)27(23-10-14-25(31)15-11-23)7-4-18-33-19-16-29(17-20-33)28(35)32-21-34(29)26-5-2-1-3-6-26/h1-3,5-6,8-15,27H,4,7,16-21H2,(H,32,35) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
ACADIA Pharmaceuticals
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 315: 1278-87 (2005)
Article DOI: 10.1124/jpet.105.092155
BindingDB Entry DOI: 10.7270/Q2Z60MNB |
More data for this
Ligand-Target Pair | |
D(4) dopamine receptor
(Homo sapiens (Human)) | BDBM26948

(8-[4,4-bis(4-fluorophenyl)butyl]-1-phenyl-1,3,8-tr...)Show SMILES Fc1ccc(cc1)C(CCCN1CCC2(CC1)N(CNC2=O)c1ccccc1)c1ccc(F)cc1 Show InChI InChI=1S/C29H31F2N3O/c30-24-12-8-22(9-13-24)27(23-10-14-25(31)15-11-23)7-4-18-33-19-16-29(17-20-33)28(35)32-21-34(29)26-5-2-1-3-6-26/h1-3,5-6,8-15,27H,4,7,16-21H2,(H,32,35) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 8.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
ACADIA Pharmaceuticals
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 315: 1278-87 (2005)
Article DOI: 10.1124/jpet.105.092155
BindingDB Entry DOI: 10.7270/Q2Z60MNB |
More data for this
Ligand-Target Pair | |
Potassium channel subfamily K member 2
(Homo sapiens (Human)) | BDBM26948

(8-[4,4-bis(4-fluorophenyl)butyl]-1-phenyl-1,3,8-tr...)Show SMILES Fc1ccc(cc1)C(CCCN1CCC2(CC1)N(CNC2=O)c1ccccc1)c1ccc(F)cc1 Show InChI InChI=1S/C29H31F2N3O/c30-24-12-8-22(9-13-24)27(23-10-14-25(31)15-11-23)7-4-18-33-19-16-29(17-20-33)28(35)32-21-34(29)26-5-2-1-3-6-26/h1-3,5-6,8-15,27H,4,7,16-21H2,(H,32,35) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 232 | n/a | n/a | 7.3 | 22 |
Korea Institute of Science and Technology
| Assay Description
The hTREK1 stable cell lines were seeded at a density of 10 000 cells/well in a 12-well plate. Whole-cell membrane currents were amplified using the ... |
Chem Biol Drug Des 88: 807-819 (2016)
Article DOI: 10.1111/cbdd.12810
BindingDB Entry DOI: 10.7270/Q2S181B1 |
More data for this
Ligand-Target Pair | |
Arachidonate 15-lipoxygenase
(Pseudomonas aeruginosa) | BDBM26948

(8-[4,4-bis(4-fluorophenyl)butyl]-1-phenyl-1,3,8-tr...)Show SMILES Fc1ccc(cc1)C(CCCN1CCC2(CC1)N(CNC2=O)c1ccccc1)c1ccc(F)cc1 Show InChI InChI=1S/C29H31F2N3O/c30-24-12-8-22(9-13-24)27(23-10-14-25(31)15-11-23)7-4-18-33-19-16-29(17-20-33)28(35)32-21-34(29)26-5-2-1-3-6-26/h1-3,5-6,8-15,27H,4,7,16-21H2,(H,32,35) | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.10E+3 | n/a | n/a | n/a | n/a | n/a | 25 |
University of California, Santa Cruz
| Assay Description
Briefly, 3 uL of enzyme (approximately 20 nM LoxA, final concentration) or buffer (no-enzyme control) was dispensed into 1536-well Greiner black clea... |
Biochemistry 55: 3329-40 (2016)
Article DOI: 10.1021/acs.biochem.6b00338
BindingDB Entry DOI: 10.7270/Q21N7ZXT |
More data for this
Ligand-Target Pair | |
Glycine receptor subunit alpha-1
(Homo sapiens (Human)) | BDBM26948

(8-[4,4-bis(4-fluorophenyl)butyl]-1-phenyl-1,3,8-tr...)Show SMILES Fc1ccc(cc1)C(CCCN1CCC2(CC1)N(CNC2=O)c1ccccc1)c1ccc(F)cc1 Show InChI InChI=1S/C29H31F2N3O/c30-24-12-8-22(9-13-24)27(23-10-14-25(31)15-11-23)7-4-18-33-19-16-29(17-20-33)28(35)32-21-34(29)26-5-2-1-3-6-26/h1-3,5-6,8-15,27H,4,7,16-21H2,(H,32,35) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a |
University of Pittsburgh
Curated by ChEMBL
| Assay Description
Potentiation of human GlyR-alpha1 expressed in Xenopus laevis oocytes assessed as induction of glycine-activated currents after 1 to 4 days by two-el... |
J Med Chem 58: 2958-66 (2015)
Article DOI: 10.1021/jm501873p
BindingDB Entry DOI: 10.7270/Q22F7Q4V |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2B
(Homo sapiens (Human)) | BDBM26948

(8-[4,4-bis(4-fluorophenyl)butyl]-1-phenyl-1,3,8-tr...)Show SMILES Fc1ccc(cc1)C(CCCN1CCC2(CC1)N(CNC2=O)c1ccccc1)c1ccc(F)cc1 Show InChI InChI=1S/C29H31F2N3O/c30-24-12-8-22(9-13-24)27(23-10-14-25(31)15-11-23)7-4-18-33-19-16-29(17-20-33)28(35)32-21-34(29)26-5-2-1-3-6-26/h1-3,5-6,8-15,27H,4,7,16-21H2,(H,32,35) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 151 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by ChEMBL
| Assay Description
Displacement of [3H]LSD from human cloned 5HT2B receptor expressed in CHO cells by liquid scintillation counting |
J Med Chem 53: 7573-86 (2010)
Article DOI: 10.1021/jm100600y
BindingDB Entry DOI: 10.7270/Q2S182RT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM26948

(8-[4,4-bis(4-fluorophenyl)butyl]-1-phenyl-1,3,8-tr...)Show SMILES Fc1ccc(cc1)C(CCCN1CCC2(CC1)N(CNC2=O)c1ccccc1)c1ccc(F)cc1 Show InChI InChI=1S/C29H31F2N3O/c30-24-12-8-22(9-13-24)27(23-10-14-25(31)15-11-23)7-4-18-33-19-16-29(17-20-33)28(35)32-21-34(29)26-5-2-1-3-6-26/h1-3,5-6,8-15,27H,4,7,16-21H2,(H,32,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibitory concentration against IKr potassium channel |
Bioorg Med Chem Lett 14: 4771-7 (2004)
Article DOI: 10.1016/j.bmcl.2004.06.070
BindingDB Entry DOI: 10.7270/Q208661K |
More data for this
Ligand-Target Pair | |
Orf1a protein
(MERS-CoV) | BDBM26948

(8-[4,4-bis(4-fluorophenyl)butyl]-1-phenyl-1,3,8-tr...)Show SMILES Fc1ccc(cc1)C(CCCN1CCC2(CC1)N(CNC2=O)c1ccccc1)c1ccc(F)cc1 Show InChI InChI=1S/C29H31F2N3O/c30-24-12-8-22(9-13-24)27(23-10-14-25(31)15-11-23)7-4-18-33-19-16-29(17-20-33)28(35)32-21-34(29)26-5-2-1-3-6-26/h1-3,5-6,8-15,27H,4,7,16-21H2,(H,32,35) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 7.47E+3 | n/a | n/a | n/a | n/a |
University of Bonn
| Assay Description
This is a review article. |
Med Res Rev (2020)
Article DOI: 10.1002/med.21724
BindingDB Entry DOI: 10.7270/Q2JS9ST6 |
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(Human SARS coronavirus (SARS-CoV) (Severe acute re...) | BDBM26948

(8-[4,4-bis(4-fluorophenyl)butyl]-1-phenyl-1,3,8-tr...)Show SMILES Fc1ccc(cc1)C(CCCN1CCC2(CC1)N(CNC2=O)c1ccccc1)c1ccc(F)cc1 Show InChI InChI=1S/C29H31F2N3O/c30-24-12-8-22(9-13-24)27(23-10-14-25(31)15-11-23)7-4-18-33-19-16-29(17-20-33)28(35)32-21-34(29)26-5-2-1-3-6-26/h1-3,5-6,8-15,27H,4,7,16-21H2,(H,32,35) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 5.96E+3 | n/a | n/a | n/a | n/a |
University of Bonn
| Assay Description
This is a review article. |
Med Res Rev (2020)
Article DOI: 10.1002/med.21724
BindingDB Entry DOI: 10.7270/Q2JS9ST6 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data