

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM27193 (1R,2S,5S,6S,9R,12S,13R,16S)-N,N,6,7,13-pentamethyl-7-azapentacyclo[10.8.0.0^{2,9}.0^{5,9}.0^{13,18}]icos-18-en-16-amine::Conessine
SMILES: [H][C@@]1(C)N(C)C[C@]23CC[C@@]4([H])[C@@]([H])(CC=C5C[C@H](CC[C@]45C)N(C)C)[C@]2([H])CC[C@]13[H]
InChI Key: InChIKey=GPLGAQQQNWMVMM-MYAJQUOBSA-N
Data: 8 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM27193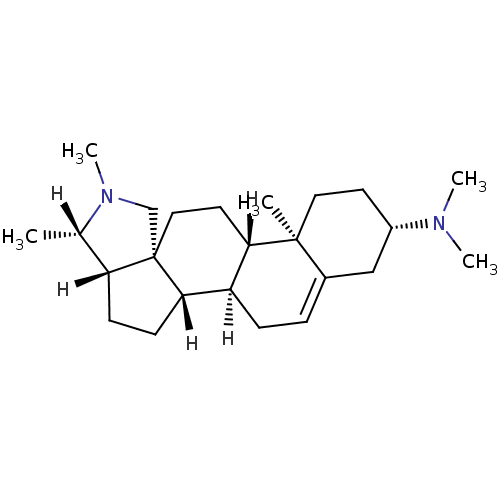 ((1R,2S,5S,6S,9R,12S,13R,16S)-N,N,6,7,13-pentamethy...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | 5.37 | -11.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Abbott Laboratories | Assay Description Competition radioligand binding assays were performed with increasing concentrations of test compound in the presence of [3H]ligand. All binding reac... | J Med Chem 51: 5423-30 (2008) Article DOI: 10.1021/jm8003625 BindingDB Entry DOI: 10.7270/Q21G0JK0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM27193 ((1R,2S,5S,6S,9R,12S,13R,16S)-N,N,6,7,13-pentamethy...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | 5.37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]N-alpha methyl histamine from human cloned histamine H3 receptor expressed in C6 cells | J Med Chem 52: 4640-9 (2009) Article DOI: 10.1021/jm900480x BindingDB Entry DOI: 10.7270/Q2Z31ZNT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM27193 ((1R,2S,5S,6S,9R,12S,13R,16S)-N,N,6,7,13-pentamethy...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | 24.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories | Assay Description Competition radioligand binding assays were performed with increasing concentrations of test compound in the presence of [3H]ligand. All binding reac... | J Med Chem 51: 5423-30 (2008) Article DOI: 10.1021/jm8003625 BindingDB Entry DOI: 10.7270/Q21G0JK0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM27193 ((1R,2S,5S,6S,9R,12S,13R,16S)-N,N,6,7,13-pentamethy...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]N-alpha methyl histamine from rat cortical membrane histamine H3 receptor | J Med Chem 52: 4640-9 (2009) Article DOI: 10.1021/jm900480x BindingDB Entry DOI: 10.7270/Q2Z31ZNT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM27193 ((1R,2S,5S,6S,9R,12S,13R,16S)-N,N,6,7,13-pentamethy...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | 66 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Arena Pharmaceuticals Curated by ChEMBL | Assay Description Displacement of N-[3H]methylhistamine from histamine H3 receptor in rat cortex | Bioorg Med Chem Lett 18: 1490-4 (2008) Article DOI: 10.1016/j.bmcl.2007.12.059 BindingDB Entry DOI: 10.7270/Q2Q81DZS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM27193 ((1R,2S,5S,6S,9R,12S,13R,16S)-N,N,6,7,13-pentamethy...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories | Assay Description Competition radioligand binding assays were performed with increasing concentrations of test compound in the presence of [3H]ligand. All binding reac... | J Med Chem 51: 5423-30 (2008) Article DOI: 10.1021/jm8003625 BindingDB Entry DOI: 10.7270/Q21G0JK0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM27193 ((1R,2S,5S,6S,9R,12S,13R,16S)-N,N,6,7,13-pentamethy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | >1.00E+4 | >-6.82 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Abbott Laboratories | Assay Description Competition radioligand binding assays were performed with increasing concentrations of test compound in the presence of [3H]ligand. All binding reac... | J Med Chem 51: 5423-30 (2008) Article DOI: 10.1021/jm8003625 BindingDB Entry DOI: 10.7270/Q21G0JK0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H2 Receptor (Homo sapiens (Human)) | BDBM27193 ((1R,2S,5S,6S,9R,12S,13R,16S)-N,N,6,7,13-pentamethy...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | >1.00E+4 | >-6.82 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Abbott Laboratories | Assay Description Competition radioligand binding assays were performed with increasing concentrations of test compound in the presence of [3H]ligand. All binding reac... | J Med Chem 51: 5423-30 (2008) Article DOI: 10.1021/jm8003625 BindingDB Entry DOI: 10.7270/Q21G0JK0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||