 Found 8 hits for monomerid = 28884
Found 8 hits for monomerid = 28884 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Anthrax Lethal Factor (LF)
(Bacillus anthracis) | BDBM28884
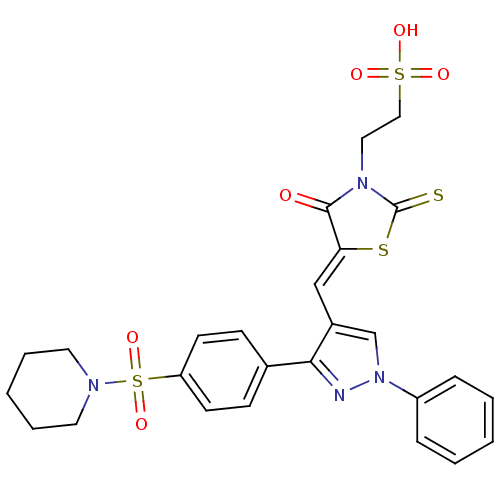
(2-[(5Z)-4-oxo-5-({1-phenyl-3-[4-(piperidine-1-sulf...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(cc2)S(=O)(=O)N2CCCCC2)-c2ccccc2)C1=O Show InChI InChI=1S/C26H26N4O6S4/c31-25-23(38-26(37)29(25)15-16-39(32,33)34)17-20-18-30(21-7-3-1-4-8-21)27-24(20)19-9-11-22(12-10-19)40(35,36)28-13-5-2-6-14-28/h1,3-4,7-12,17-18H,2,5-6,13-16H2,(H,32,33,34)/b23-17- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.07E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Burnham Institute for Medical Research
| Assay Description
The fluorescence peptide cleavage assay was performed in a 96-well plate in which each reaction mixture contained MAPKKide, LF (List Biological Labor... |
Chem Biol Drug Des 71: 131-9 (2008)
Article DOI: 10.1111/j.1747-0285.2007.00617.x
BindingDB Entry DOI: 10.7270/Q2KW5DCN |
More data for this
Ligand-Target Pair | |
Botulinum Neurotoxin Type A
(Clostridium botulinum) | BDBM28884

(2-[(5Z)-4-oxo-5-({1-phenyl-3-[4-(piperidine-1-sulf...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(cc2)S(=O)(=O)N2CCCCC2)-c2ccccc2)C1=O Show InChI InChI=1S/C26H26N4O6S4/c31-25-23(38-26(37)29(25)15-16-39(32,33)34)17-20-18-30(21-7-3-1-4-8-21)27-24(20)19-9-11-22(12-10-19)40(35,36)28-13-5-2-6-14-28/h1,3-4,7-12,17-18H,2,5-6,13-16H2,(H,32,33,34)/b23-17- | PDB
MMDB
KEGG
B.MOAD
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.01E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Burnham Institute for Medical Research
| Assay Description
The fluorescence peptide cleavage assay was performed in a 96-well plates in which each reaction mixture contained SNAPtide, BoNT /A (List Biological... |
Chem Biol Drug Des 71: 131-9 (2008)
Article DOI: 10.1111/j.1747-0285.2007.00617.x
BindingDB Entry DOI: 10.7270/Q2KW5DCN |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase (VHR)
(Homo sapiens (Human)) | BDBM28884

(2-[(5Z)-4-oxo-5-({1-phenyl-3-[4-(piperidine-1-sulf...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(cc2)S(=O)(=O)N2CCCCC2)-c2ccccc2)C1=O Show InChI InChI=1S/C26H26N4O6S4/c31-25-23(38-26(37)29(25)15-16-39(32,33)34)17-20-18-30(21-7-3-1-4-8-21)27-24(20)19-9-11-22(12-10-19)40(35,36)28-13-5-2-6-14-28/h1,3-4,7-12,17-18H,2,5-6,13-16H2,(H,32,33,34)/b23-17- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 270 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant vaccina H1-related phosphatase |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
phosphatase Cdc25
(Homo sapiens (Human)) | BDBM28884

(2-[(5Z)-4-oxo-5-({1-phenyl-3-[4-(piperidine-1-sulf...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(cc2)S(=O)(=O)N2CCCCC2)-c2ccccc2)C1=O Show InChI InChI=1S/C26H26N4O6S4/c31-25-23(38-26(37)29(25)15-16-39(32,33)34)17-20-18-30(21-7-3-1-4-8-21)27-24(20)19-9-11-22(12-10-19)40(35,36)28-13-5-2-6-14-28/h1,3-4,7-12,17-18H,2,5-6,13-16H2,(H,32,33,34)/b23-17- | MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Cdc25a |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Protein-tyrosine phosphatase 1B
(Homo sapiens (Human)) | BDBM28884

(2-[(5Z)-4-oxo-5-({1-phenyl-3-[4-(piperidine-1-sulf...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(cc2)S(=O)(=O)N2CCCCC2)-c2ccccc2)C1=O Show InChI InChI=1S/C26H26N4O6S4/c31-25-23(38-26(37)29(25)15-16-39(32,33)34)17-20-18-30(21-7-3-1-4-8-21)27-24(20)19-9-11-22(12-10-19)40(35,36)28-13-5-2-6-14-28/h1,3-4,7-12,17-18H,2,5-6,13-16H2,(H,32,33,34)/b23-17- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant PTP1B |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Hematopoietic protein-tyrosine phosphatase (HEPTP)
(Homo sapiens (Human)) | BDBM28884

(2-[(5Z)-4-oxo-5-({1-phenyl-3-[4-(piperidine-1-sulf...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(cc2)S(=O)(=O)N2CCCCC2)-c2ccccc2)C1=O Show InChI InChI=1S/C26H26N4O6S4/c31-25-23(38-26(37)29(25)15-16-39(32,33)34)17-20-18-30(21-7-3-1-4-8-21)27-24(20)19-9-11-22(12-10-19)40(35,36)28-13-5-2-6-14-28/h1,3-4,7-12,17-18H,2,5-6,13-16H2,(H,32,33,34)/b23-17- | PDB
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant HePTP |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Dual Specificity Protein Phosphatase 1
(Homo sapiens (Human)) | BDBM28884

(2-[(5Z)-4-oxo-5-({1-phenyl-3-[4-(piperidine-1-sulf...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(cc2)S(=O)(=O)N2CCCCC2)-c2ccccc2)C1=O Show InChI InChI=1S/C26H26N4O6S4/c31-25-23(38-26(37)29(25)15-16-39(32,33)34)17-20-18-30(21-7-3-1-4-8-21)27-24(20)19-9-11-22(12-10-19)40(35,36)28-13-5-2-6-14-28/h1,3-4,7-12,17-18H,2,5-6,13-16H2,(H,32,33,34)/b23-17- | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant MKP1 |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |
Leukocyte common antigen
(Homo sapiens (Human)) | BDBM28884

(2-[(5Z)-4-oxo-5-({1-phenyl-3-[4-(piperidine-1-sulf...)Show SMILES OS(=O)(=O)CCN1C(=S)S\C(=C/c2cn(nc2-c2ccc(cc2)S(=O)(=O)N2CCCCC2)-c2ccccc2)C1=O Show InChI InChI=1S/C26H26N4O6S4/c31-25-23(38-26(37)29(25)15-16-39(32,33)34)17-20-18-30(21-7-3-1-4-8-21)27-24(20)19-9-11-22(12-10-19)40(35,36)28-13-5-2-6-14-28/h1,3-4,7-12,17-18H,2,5-6,13-16H2,(H,32,33,34)/b23-17- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Medical Research
Curated by ChEMBL
| Assay Description
Inhibition of recombinant CD45 |
J Med Chem 52: 6716-23 (2009)
Article DOI: 10.1021/jm901016k
BindingDB Entry DOI: 10.7270/Q2VX0GK3 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data