

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM294045 US10106571, Example 2::US10106571, Example 23
SMILES: CC(C)OC(=O)[C@H](C)NP(=O)(OC[C@H]1O[C@@H](n2ccc(=O)[nH]c2=O)[C@@](F)(Cl)[C@@H]1O)Oc1ccccc1
InChI Key:
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| RNA polymerase (NS5B) (Hepatitis C virus (HCV)) | BDBM294045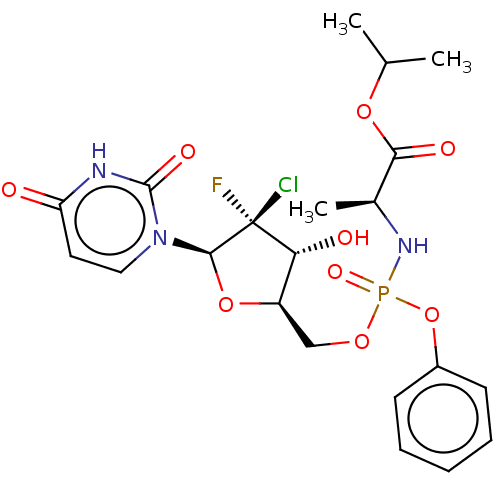 (US10106571, Example 2 | US10106571, Example 23) | PDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | n/a | n/a | 55 | n/a | n/a | n/a | n/a |
MEDIVIR AB US Patent | Assay Description The assay utilizes the stably transfected cell line Huh-7 luc/neo (hereafter referred to as Huh-Luc). This cell line harbors an RNA encoding a bicist... | US Patent US10106571 (2018) BindingDB Entry DOI: 10.7270/Q2NP26GX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| RNA polymerase (NS5B) (Hepatitis C virus (HCV)) | BDBM294045 (US10106571, Example 2 | US10106571, Example 23) | PDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | n/a | n/a | 5.10E+3 | n/a | n/a | n/a | n/a |
MEDIVIR AB US Patent | Assay Description The assay utilizes the stably transfected cell line Huh-7 luc/neo (hereafter referred to as Huh-Luc). This cell line harbors an RNA encoding a bicist... | US Patent US10106571 (2018) BindingDB Entry DOI: 10.7270/Q2NP26GX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D6 (Homo sapiens (Human)) | BDBM294045 (US10106571, Example 2 | US10106571, Example 23) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine Curated by ChEMBL | Assay Description Inhibition of human CYP2D6 using quinidine as substrate | J Med Chem 60: 5424-5437 (2017) Article DOI: 10.1021/acs.jmedchem.7b00067 BindingDB Entry DOI: 10.7270/Q2BZ6873 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2C9 (Homo sapiens (Human)) | BDBM294045 (US10106571, Example 2 | US10106571, Example 23) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine Curated by ChEMBL | Assay Description Inhibition of human CYP2C9 using sulfaphenazole as substrate | J Med Chem 60: 5424-5437 (2017) Article DOI: 10.1021/acs.jmedchem.7b00067 BindingDB Entry DOI: 10.7270/Q2BZ6873 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 1A2 (Homo sapiens (Human)) | BDBM294045 (US10106571, Example 2 | US10106571, Example 23) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine Curated by ChEMBL | Assay Description Inhibition of human CYP1A2 using alpha-naphthoflavone as substrate | J Med Chem 60: 5424-5437 (2017) Article DOI: 10.1021/acs.jmedchem.7b00067 BindingDB Entry DOI: 10.7270/Q2BZ6873 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 3A4 (Homo sapiens (Human)) | BDBM294045 (US10106571, Example 2 | US10106571, Example 23) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 6.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine Curated by ChEMBL | Assay Description Inhibition of human CYP3A4 using ketoconazole as substrate | J Med Chem 60: 5424-5437 (2017) Article DOI: 10.1021/acs.jmedchem.7b00067 BindingDB Entry DOI: 10.7270/Q2BZ6873 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||