

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM29744 hydroxyethylene tripeptide inhibitor, 1
SMILES: CCCCNC(=O)[C@H](C)C[C@H](O)[C@H](CC(C)C)NC(=O)[C@H](CCSC)NC(=O)CCC(C)C
InChI Key: InChIKey=XTRMRNMVFXWTCC-MLNNCEHLSA-N
Data: 5 IC50
PDB links: 2 PDB IDs match this monomer. 1 PDB ID contains inhibitors having a similarity of 90% to this monomer.
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| beta-Secretase (BACE-1) (Homo sapiens (Human)) | BDBM29744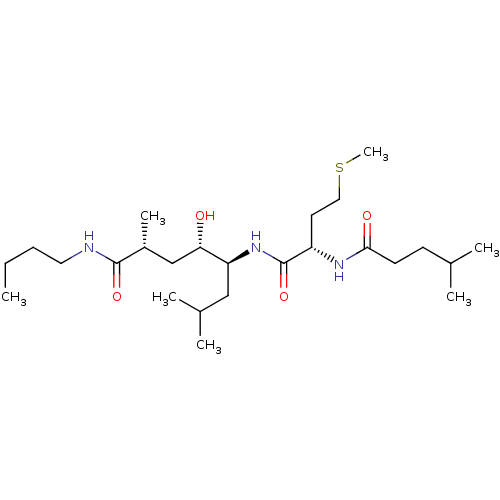 (hydroxyethylene tripeptide inhibitor, 1) | PDB MMDB KEGG B.MOAD DrugBank GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | 4.5 | 22 |
Novartis | Assay Description Inhibition assays were done in black 96-well plates by adding test compounds to the respective enzyme in assay buffer. Plates were incubated at room ... | Bioorg Med Chem Lett 19: 1361-5 (2009) Article DOI: 10.1016/j.bmcl.2009.01.036 BindingDB Entry DOI: 10.7270/Q2JS9NS3 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Cathepsin D (Homo sapiens (Human)) | BDBM29744 (hydroxyethylene tripeptide inhibitor, 1) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 370 | n/a | n/a | n/a | n/a | 3.1 | 22 |
Novartis | Assay Description Inhibition assays were done in black 96-well plates by adding test compounds to the respective enzyme in assay buffer. Plates were incubated at room ... | Bioorg Med Chem Lett 19: 1361-5 (2009) Article DOI: 10.1016/j.bmcl.2009.01.036 BindingDB Entry DOI: 10.7270/Q2JS9NS3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cathepsin D (Homo sapiens (Human)) | BDBM29744 (hydroxyethylene tripeptide inhibitor, 1) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 370 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research Curated by ChEMBL | Assay Description Inhibition of human cathepsin D | Bioorg Med Chem Lett 19: 1366-70 (2009) Article DOI: 10.1016/j.bmcl.2009.01.055 BindingDB Entry DOI: 10.7270/Q2SB45S3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM29744 (hydroxyethylene tripeptide inhibitor, 1) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research Curated by ChEMBL | Assay Description Inhibition of BACE1 expressed in CHO cells expressing human recombinant APP assessed as amyloid beta40 aggregation | Bioorg Med Chem Lett 19: 1366-70 (2009) Article DOI: 10.1016/j.bmcl.2009.01.055 BindingDB Entry DOI: 10.7270/Q2SB45S3 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM29744 (hydroxyethylene tripeptide inhibitor, 1) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research Curated by ChEMBL | Assay Description Inhibition of BACE1 | Bioorg Med Chem Lett 19: 1366-70 (2009) Article DOI: 10.1016/j.bmcl.2009.01.055 BindingDB Entry DOI: 10.7270/Q2SB45S3 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||