

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM29781 6,6,5 tricyclic sulfonamide, 21
SMILES: CCc1cn2CS(=O)(=O)N(C)c3cc(cc1c23)C(=O)N[C@@H](Cc1ccccc1)[C@H](O)CNCc1cccc(OC)c1
InChI Key: InChIKey=BIFXFADRRVXKMW-LMSSTIIKSA-N
Data: 3 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM29781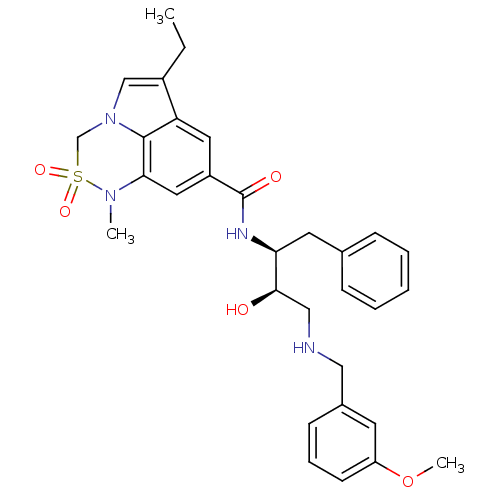 (6,6,5 tricyclic sulfonamide, 21) | PDB MMDB KEGG B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 8 | n/a | n/a | n/a | n/a | 4.5 | 22 |
GSK | Assay Description Enzyme activity and inhibition were assayed using a fluorescent FAM-[SEVNLDAEFK]-TAMRA substrate. Control reactions with no enzyme were included in e... | Bioorg Med Chem Lett 19: 3669-73 (2009) Article DOI: 10.1016/j.bmcl.2009.03.150 BindingDB Entry DOI: 10.7270/Q29885BR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cathepsin D (Homo sapiens (Human)) | BDBM29781 (6,6,5 tricyclic sulfonamide, 21) | PDB MMDB NCI pathway Reactome pathway KEGG B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GSK | Assay Description Enzyme activity and inhibition were assayed using a fluorescent FAM-[SEVNLDAEFK]-TAMRA substrate. Control reactions with no enzyme were included in e... | Bioorg Med Chem Lett 19: 3669-73 (2009) Article DOI: 10.1016/j.bmcl.2009.03.150 BindingDB Entry DOI: 10.7270/Q29885BR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 2 (Homo sapiens (Human)) | BDBM29781 (6,6,5 tricyclic sulfonamide, 21) | PDB GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 350 | n/a | n/a | n/a | n/a | n/a | n/a |
GSK | Assay Description Enzyme activity and inhibition were assayed using a fluorescent FAM-[SEVNLDAEFK]-TAMRA substrate. Control reactions with no enzyme were included in e... | Bioorg Med Chem Lett 19: 3669-73 (2009) Article DOI: 10.1016/j.bmcl.2009.03.150 BindingDB Entry DOI: 10.7270/Q29885BR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||