 Found 10 hits for monomerid = 30185
Found 10 hits for monomerid = 30185 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
MAP kinase-activated protein kinase 2
(Homo sapiens (Human)) | BDBM30185
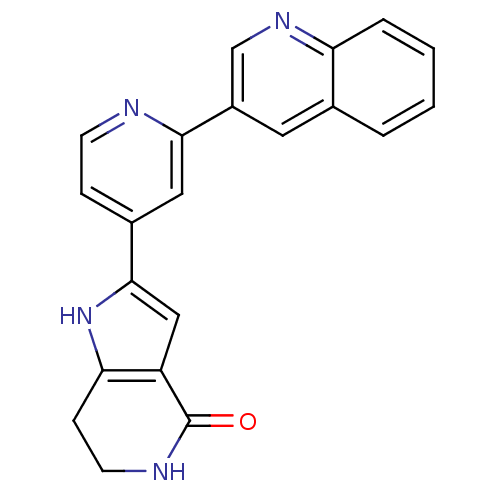
(CHEMBL226403 | Pyrrolopyridine, 16)Show SMILES O=C1NCCc2[nH]c(cc12)-c1ccnc(c1)-c1cnc2ccccc2c1 Show InChI InChI=1S/C21H16N4O/c26-21-16-11-20(25-18(16)6-8-23-21)14-5-7-22-19(10-14)15-9-13-3-1-2-4-17(13)24-12-15/h1-5,7,9-12,25H,6,8H2,(H,23,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 8.5 | n/a | n/a | n/a | n/a | 7.5 | 30 |
Pfizer
| Assay Description
The phosphorylation of HSP27 peptide by MAPKAPK2 was measured using an anion exchange resin capture assay method. The reaction was carried out in rea... |
J Med Chem 50: 2647-54 (2007)
Article DOI: 10.1021/jm0611004
BindingDB Entry DOI: 10.7270/Q2794313 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
MAP kinase-activated protein kinase 3
(Homo sapiens (Human)) | BDBM30185

(CHEMBL226403 | Pyrrolopyridine, 16)Show SMILES O=C1NCCc2[nH]c(cc12)-c1ccnc(c1)-c1cnc2ccccc2c1 Show InChI InChI=1S/C21H16N4O/c26-21-16-11-20(25-18(16)6-8-23-21)14-5-7-22-19(10-14)15-9-13-3-1-2-4-17(13)24-12-15/h1-5,7,9-12,25H,6,8H2,(H,23,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 210 | n/a | n/a | n/a | n/a | 7.5 | 30 |
Pfizer
| Assay Description
Compounds were evaluated as inhibitors of kinase by measuring their effect on kinase induced phosphorylation of substrate. IC50 values were reported ... |
J Med Chem 50: 2647-54 (2007)
Article DOI: 10.1021/jm0611004
BindingDB Entry DOI: 10.7270/Q2794313 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
MAP kinase-activated protein kinase 5
(Homo sapiens (Human)) | BDBM30185

(CHEMBL226403 | Pyrrolopyridine, 16)Show SMILES O=C1NCCc2[nH]c(cc12)-c1ccnc(c1)-c1cnc2ccccc2c1 Show InChI InChI=1S/C21H16N4O/c26-21-16-11-20(25-18(16)6-8-23-21)14-5-7-22-19(10-14)15-9-13-3-1-2-4-17(13)24-12-15/h1-5,7,9-12,25H,6,8H2,(H,23,26) | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 81 | n/a | n/a | n/a | n/a | 7.5 | 30 |
Pfizer
| Assay Description
Compounds were evaluated as inhibitors of kinase by measuring their effect on kinase induced phosphorylation of substrate. IC50 values were reported ... |
J Med Chem 50: 2647-54 (2007)
Article DOI: 10.1021/jm0611004
BindingDB Entry DOI: 10.7270/Q2794313 |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 1
(Homo sapiens (Human)) | BDBM30185

(CHEMBL226403 | Pyrrolopyridine, 16)Show SMILES O=C1NCCc2[nH]c(cc12)-c1ccnc(c1)-c1cnc2ccccc2c1 Show InChI InChI=1S/C21H16N4O/c26-21-16-11-20(25-18(16)6-8-23-21)14-5-7-22-19(10-14)15-9-13-3-1-2-4-17(13)24-12-15/h1-5,7,9-12,25H,6,8H2,(H,23,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.44E+3 | n/a | n/a | n/a | n/a | 7.5 | 23 |
Pfizer
| Assay Description
Compounds were evaluated as inhibitors of kinase by measuring their effect on kinase induced phosphorylation of substrate. IC50 values were reported ... |
J Med Chem 50: 2647-54 (2007)
Article DOI: 10.1021/jm0611004
BindingDB Entry DOI: 10.7270/Q2794313 |
More data for this
Ligand-Target Pair | |
MAP kinase-activated protein kinase 5
(Homo sapiens (Human)) | BDBM30185

(CHEMBL226403 | Pyrrolopyridine, 16)Show SMILES O=C1NCCc2[nH]c(cc12)-c1ccnc(c1)-c1cnc2ccccc2c1 Show InChI InChI=1S/C21H16N4O/c26-21-16-11-20(25-18(16)6-8-23-21)14-5-7-22-19(10-14)15-9-13-3-1-2-4-17(13)24-12-15/h1-5,7,9-12,25H,6,8H2,(H,23,26) | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of MAPKAPK5 (unknown origin) |
J Med Chem 56: 6991-7002 (2013)
Article DOI: 10.1021/jm400798j
BindingDB Entry DOI: 10.7270/Q2G44RQC |
More data for this
Ligand-Target Pair | |
Ribosomal protein S6 kinase alpha 5
(Homo sapiens (Human)) | BDBM30185

(CHEMBL226403 | Pyrrolopyridine, 16)Show SMILES O=C1NCCc2[nH]c(cc12)-c1ccnc(c1)-c1cnc2ccccc2c1 Show InChI InChI=1S/C21H16N4O/c26-21-16-11-20(25-18(16)6-8-23-21)14-5-7-22-19(10-14)15-9-13-3-1-2-4-17(13)24-12-15/h1-5,7,9-12,25H,6,8H2,(H,23,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer
| Assay Description
Compounds were evaluated as inhibitors of kinase by measuring their effect on kinase induced phosphorylation of substrate. IC50 values were reported ... |
J Med Chem 50: 2647-54 (2007)
Article DOI: 10.1021/jm0611004
BindingDB Entry DOI: 10.7270/Q2794313 |
More data for this
Ligand-Target Pair | |
Ribosomal protein S6 kinase alpha 4
(Homo sapiens (Human)) | BDBM30185

(CHEMBL226403 | Pyrrolopyridine, 16)Show SMILES O=C1NCCc2[nH]c(cc12)-c1ccnc(c1)-c1cnc2ccccc2c1 Show InChI InChI=1S/C21H16N4O/c26-21-16-11-20(25-18(16)6-8-23-21)14-5-7-22-19(10-14)15-9-13-3-1-2-4-17(13)24-12-15/h1-5,7,9-12,25H,6,8H2,(H,23,26) | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer
| Assay Description
Compounds were evaluated as inhibitors of kinase by measuring their effect on kinase induced phosphorylation of substrate. IC50 values were reported ... |
J Med Chem 50: 2647-54 (2007)
Article DOI: 10.1021/jm0611004
BindingDB Entry DOI: 10.7270/Q2794313 |
More data for this
Ligand-Target Pair | |
MAP kinase-activated protein kinase 2
(Homo sapiens (Human)) | BDBM30185

(CHEMBL226403 | Pyrrolopyridine, 16)Show SMILES O=C1NCCc2[nH]c(cc12)-c1ccnc(c1)-c1cnc2ccccc2c1 Show InChI InChI=1S/C21H16N4O/c26-21-16-11-20(25-18(16)6-8-23-21)14-5-7-22-19(10-14)15-9-13-3-1-2-4-17(13)24-12-15/h1-5,7,9-12,25H,6,8H2,(H,23,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 8.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Princ
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant MK2 expressed in Escherichia coli BL21(DE3) after 60 mins |
J Med Chem 51: 6225-9 (2008)
Article DOI: 10.1021/jm800747w
BindingDB Entry DOI: 10.7270/Q2JM29GJ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
MAP kinase-activated protein kinase 2
(Homo sapiens (Human)) | BDBM30185

(CHEMBL226403 | Pyrrolopyridine, 16)Show SMILES O=C1NCCc2[nH]c(cc12)-c1ccnc(c1)-c1cnc2ccccc2c1 Show InChI InChI=1S/C21H16N4O/c26-21-16-11-20(25-18(16)6-8-23-21)14-5-7-22-19(10-14)15-9-13-3-1-2-4-17(13)24-12-15/h1-5,7,9-12,25H,6,8H2,(H,23,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 49 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of MK2 pretreated for 30 mins before substrate addition measured after 2 hrs by IMAP assay |
Bioorg Med Chem Lett 21: 3818-22 (2011)
Article DOI: 10.1016/j.bmcl.2011.04.018
BindingDB Entry DOI: 10.7270/Q26973W4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
MAP kinase-interacting serine/threonine-protein kinase 1
(Homo sapiens (Human)) | BDBM30185

(CHEMBL226403 | Pyrrolopyridine, 16)Show SMILES O=C1NCCc2[nH]c(cc12)-c1ccnc(c1)-c1cnc2ccccc2c1 Show InChI InChI=1S/C21H16N4O/c26-21-16-11-20(25-18(16)6-8-23-21)14-5-7-22-19(10-14)15-9-13-3-1-2-4-17(13)24-12-15/h1-5,7,9-12,25H,6,8H2,(H,23,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer
| Assay Description
Compounds were evaluated as inhibitors of kinase by measuring their effect on kinase induced phosphorylation of substrate. IC50 values were reported ... |
J Med Chem 50: 2647-54 (2007)
Article DOI: 10.1021/jm0611004
BindingDB Entry DOI: 10.7270/Q2794313 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data