

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM3103 2-aminopyrido[2,3-d]pyrimidin-7(8H)-one 70::3-[6-(2,6-Dichlorophenyl)-8-methyl-7-oxo-7,8-dihydropyrido[2,3-d]pyrimidin-2-ylamino]benzoic Acid::3-{[6-(2,6-dichlorophenyl)-8-methyl-7-oxo-7H,8H-pyrido[2,3-d]pyrimidin-2-yl]amino}benzoic acid::PD 166285 analog
SMILES: Cn1c2nc(Nc3cccc(c3)C(O)=O)ncc2cc(-c2c(Cl)cccc2Cl)c1=O
InChI Key: InChIKey=RBURTLAMOSHNGX-UHFFFAOYSA-N
Data: 4 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Platelet-derived growth factor receptor beta (Homo sapiens (Human)) | BDBM3103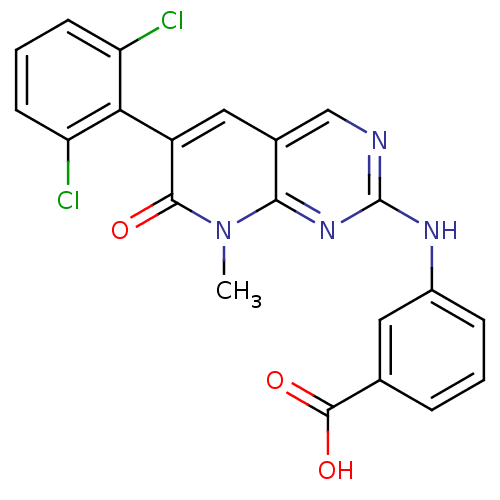 (2-aminopyrido[2,3-d]pyrimidin-7(8H)-one 70 | 3-[6-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
Parke-Davis Pharmaceutical Research | Assay Description IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the transfer of the terminal phosphate from [gamma-32P] labe... | J Med Chem 41: 3276-92 (1998) Article DOI: 10.1021/jm9802259 BindingDB Entry DOI: 10.7270/Q27P8WK3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM3103 (2-aminopyrido[2,3-d]pyrimidin-7(8H)-one 70 | 3-[6-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 190 | n/a | n/a | n/a | n/a | n/a | n/a |
Parke-Davis Pharmaceutical Research | Assay Description IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the transfer of the terminal phosphate from [gamma-32P] labe... | J Med Chem 41: 3276-92 (1998) Article DOI: 10.1021/jm9802259 BindingDB Entry DOI: 10.7270/Q27P8WK3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proto-oncogene tyrosine-protein kinase Src (Homo sapiens (Human)) | BDBM3103 (2-aminopyrido[2,3-d]pyrimidin-7(8H)-one 70 | 3-[6-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Parke-Davis Pharmaceutical Research | Assay Description IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the transfer of the terminal phosphate from [gamma-32P] labe... | J Med Chem 41: 3276-92 (1998) Article DOI: 10.1021/jm9802259 BindingDB Entry DOI: 10.7270/Q27P8WK3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fibroblast growth factor receptor 1 (Homo sapiens (Human)) | BDBM3103 (2-aminopyrido[2,3-d]pyrimidin-7(8H)-one 70 | 3-[6-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
Parke-Davis Pharmaceutical Research | Assay Description IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the transfer of the terminal phosphate from [gamma-32P] labe... | J Med Chem 41: 3276-92 (1998) Article DOI: 10.1021/jm9802259 BindingDB Entry DOI: 10.7270/Q27P8WK3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||