

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
null
SMILES: CC(C)[NH2+]C[C@@H](O)COc1cccc2ccccc12
InChI Key: InChIKey=AQHHHDLHHXJYJD-CQSZACIVSA-O
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-hydroxytryptamine receptor 1A (Rattus norvegicus (rat)) | BDBM36108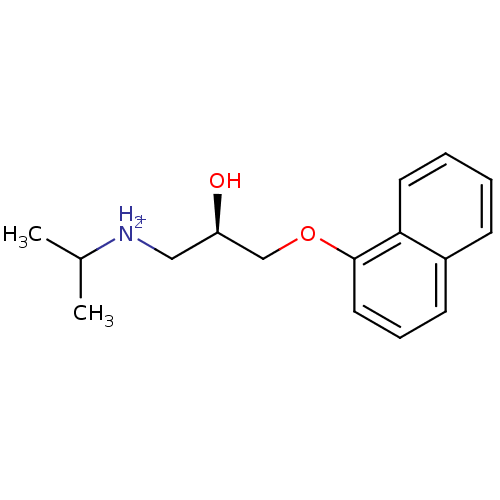 ((R)-propranolol) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | PubMed | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibitory activity against 5-hydroxytryptamine 1A receptor of rat hippocampal tissue using [3H]OH-DPAT as radioligand. | J Med Chem 32: 859-63 (1989) BindingDB Entry DOI: 10.7270/Q2571CMK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ATP-dependent translocase ABCB1 (Homo sapiens (Human)) | BDBM36108 ((R)-propranolol) | PDB UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 5.83E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Bulgarian Academy of Sciences Curated by ChEMBL | Assay Description Concentration required for 50% inhibition at binding site of human P-Glycoprotein (P-gp) in one-affinity model | J Med Chem 45: 5671-86 (2002) BindingDB Entry DOI: 10.7270/Q23B60VG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 1A (Rattus norvegicus (rat)) | BDBM36108 ((R)-propranolol) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 170 | n/a | n/a | n/a | n/a | n/a | n/a |
Merrell Dow Research Institute Curated by ChEMBL | Assay Description Binding affinity towards 5-hydroxytryptamine 1A receptor in rat frontal cortex using [3H]8-OH-DPAT as a radioligand | J Med Chem 31: 1087-93 (1988) BindingDB Entry DOI: 10.7270/Q20Z74K6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 1B (Rattus norvegicus (Rat)) | BDBM36108 ((R)-propranolol) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 490 | n/a | n/a | n/a | n/a | n/a | n/a |
Merrell Dow Research Institute Curated by ChEMBL | Assay Description Binding affinity against 5-HT1B receptor in rat frontal cortex using [3H]-5-HT in presence of 0.1 uM [3H]-8-OH-DPAT as a radioligand | J Med Chem 31: 1087-93 (1988) BindingDB Entry DOI: 10.7270/Q20Z74K6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serotonin 2 (5-HT2) receptor (RAT-Rattus norvegicus (Rat)-Rattus norvegicus (rat...) | BDBM36108 ((R)-propranolol) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 7.94E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merrell Dow Research Institute Curated by ChEMBL | Assay Description Binding affinity towards 5-hydroxytryptamine 2 receptor in rat frontal cortex using [3H]ketanserin as radioligand | J Med Chem 31: 1087-93 (1988) BindingDB Entry DOI: 10.7270/Q20Z74K6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D26 (Rattus norvegicus) | BDBM36108 ((R)-propranolol) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 420 | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Curated by ChEMBL | Assay Description Inhibition of MAMC O-dealkylation mediated by rat Cytochrome P450 2D2 expressed in Saccharomyces cerevisiae | J Med Chem 46: 74-86 (2002) Article DOI: 10.1021/jm0209578 BindingDB Entry DOI: 10.7270/Q2K64JSD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D1 (Rattus norvegicus) | BDBM36108 ((R)-propranolol) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.82E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Curated by ChEMBL | Assay Description Inhibition of MAMC O-dealkylation mediated by rat Cytochrome P450 2D1 expressed in Saccharomyces cerevisiae | J Med Chem 46: 74-86 (2002) Article DOI: 10.1021/jm0209578 BindingDB Entry DOI: 10.7270/Q2K64JSD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D6 (Homo sapiens (Human)) | BDBM36108 ((R)-propranolol) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Curated by ChEMBL | Assay Description Inhibition of MAMC O-dealkylation mediated by human Cytochrome P450 2D6 expressed in human lymphoblastoid cell line | J Med Chem 46: 74-86 (2002) Article DOI: 10.1021/jm0209578 BindingDB Entry DOI: 10.7270/Q2K64JSD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D3 (Rattus norvegicus) | BDBM36108 ((R)-propranolol) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.55E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Curated by ChEMBL | Assay Description Inhibition of MAMC O-dealkylation mediated by rat Cytochrome P450 2D3 expressed in Saccharomyces cerevisiae | J Med Chem 46: 74-86 (2002) Article DOI: 10.1021/jm0209578 BindingDB Entry DOI: 10.7270/Q2K64JSD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D4 (Rattus norvegicus) | BDBM36108 ((R)-propranolol) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.17E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Curated by ChEMBL | Assay Description Inhibition of MAMC O-dealkylation mediated by rat Cytochrome P450 2D4 expressed in Saccharomyces cerevisiae | J Med Chem 46: 74-86 (2002) Article DOI: 10.1021/jm0209578 BindingDB Entry DOI: 10.7270/Q2K64JSD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cell (A) | Syringe (B) | Cell Links | Syringe Links | Cell + Syr Links | ΔG° kcal/mole | -TΔS° kcal/mole | ΔH° kcal/mole | log K | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|
BDBM11 | BDBM36108 | CHEBI KEGG MMDB PC cid PC sid PDB | CHEBI KEGG PC cid PC sid | -2.82 | 2.24 | -5.06 | 2.06 | 4.80 | 25 | |
Japan Science and Technology Agency | J Am Chem Soc 122: 4418-35 (2000) | |||||||||