 Found 17 hits for monomerid = 50004918
Found 17 hits for monomerid = 50004918 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM50004918
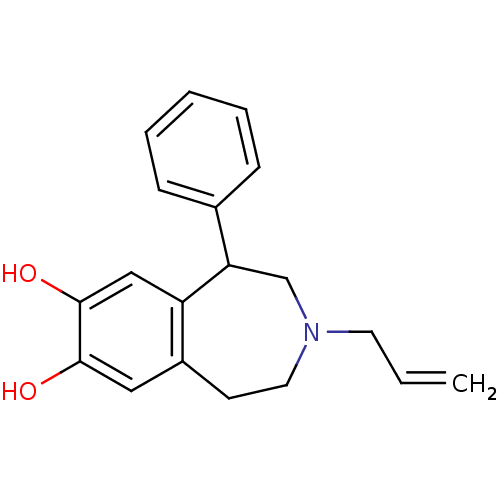
((+/-)-APD3-Allyl-1-phenyl-2,3,4,5-tetrahydro-1H-be...)Show InChI InChI=1S/C19H21NO2/c1-2-9-20-10-8-15-11-18(21)19(22)12-16(15)17(13-20)14-6-4-3-5-7-14/h2-7,11-12,17,21-22H,1,8-10,13H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SRI International
Curated by PDSP Ki Database
| |
NIDA Res Monogr 178: 440-66 (1998)
BindingDB Entry DOI: 10.7270/Q23J3BH2 |
More data for this
Ligand-Target Pair | |
D(1A) dopamine receptor
(RAT) | BDBM50004918

((+/-)-APD3-Allyl-1-phenyl-2,3,4,5-tetrahydro-1H-be...)Show InChI InChI=1S/C19H21NO2/c1-2-9-20-10-8-15-11-18(21)19(22)12-16(15)17(13-20)14-6-4-3-5-7-14/h2-7,11-12,17,21-22H,1,8-10,13H2 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 10.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard University
Curated by PDSP Ki Database
| |
Eur J Pharmacol 474: 137-40 (2003)
Article DOI: 10.1016/s0014-2999(03)02008-9
BindingDB Entry DOI: 10.7270/Q2DN43MQ |
More data for this
Ligand-Target Pair | |
D(1A) dopamine receptor
(Homo sapiens (Human)) | BDBM50004918

((+/-)-APD3-Allyl-1-phenyl-2,3,4,5-tetrahydro-1H-be...)Show InChI InChI=1S/C19H21NO2/c1-2-9-20-10-8-15-11-18(21)19(22)12-16(15)17(13-20)14-6-4-3-5-7-14/h2-7,11-12,17,21-22H,1,8-10,13H2 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northeastern University
Curated by ChEMBL
| Assay Description
The compound was evaluated for the dissociation constant for inhibiting the binding of [3H]-SCH- 23390 at Dopamine receptor D1 |
J Med Chem 34: 3366-71 (1992)
BindingDB Entry DOI: 10.7270/Q26H4J17 |
More data for this
Ligand-Target Pair | |
D(1A) dopamine receptor
(Homo sapiens (Human)) | BDBM50004918

((+/-)-APD3-Allyl-1-phenyl-2,3,4,5-tetrahydro-1H-be...)Show InChI InChI=1S/C19H21NO2/c1-2-9-20-10-8-15-11-18(21)19(22)12-16(15)17(13-20)14-6-4-3-5-7-14/h2-7,11-12,17,21-22H,1,8-10,13H2 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 73 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SRI International
Curated by PDSP Ki Database
| |
NIDA Res Monogr 178: 440-66 (1998)
BindingDB Entry DOI: 10.7270/Q23J3BH2 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50004918

((+/-)-APD3-Allyl-1-phenyl-2,3,4,5-tetrahydro-1H-be...)Show InChI InChI=1S/C19H21NO2/c1-2-9-20-10-8-15-11-18(21)19(22)12-16(15)17(13-20)14-6-4-3-5-7-14/h2-7,11-12,17,21-22H,1,8-10,13H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 580 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SRI International
Curated by PDSP Ki Database
| |
NIDA Res Monogr 178: 440-66 (1998)
BindingDB Entry DOI: 10.7270/Q23J3BH2 |
More data for this
Ligand-Target Pair | |
adrenergic Alpha2
(RAT) | BDBM50004918

((+/-)-APD3-Allyl-1-phenyl-2,3,4,5-tetrahydro-1H-be...)Show InChI InChI=1S/C19H21NO2/c1-2-9-20-10-8-15-11-18(21)19(22)12-16(15)17(13-20)14-6-4-3-5-7-14/h2-7,11-12,17,21-22H,1,8-10,13H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 632 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard University
Curated by PDSP Ki Database
| |
Eur J Pharmacol 474: 137-40 (2003)
Article DOI: 10.1016/s0014-2999(03)02008-9
BindingDB Entry DOI: 10.7270/Q2DN43MQ |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Rattus norvegicus (rat)) | BDBM50004918

((+/-)-APD3-Allyl-1-phenyl-2,3,4,5-tetrahydro-1H-be...)Show InChI InChI=1S/C19H21NO2/c1-2-9-20-10-8-15-11-18(21)19(22)12-16(15)17(13-20)14-6-4-3-5-7-14/h2-7,11-12,17,21-22H,1,8-10,13H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard University
Curated by PDSP Ki Database
| |
Eur J Pharmacol 474: 137-40 (2003)
Article DOI: 10.1016/s0014-2999(03)02008-9
BindingDB Entry DOI: 10.7270/Q2DN43MQ |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50004918

((+/-)-APD3-Allyl-1-phenyl-2,3,4,5-tetrahydro-1H-be...)Show InChI InChI=1S/C19H21NO2/c1-2-9-20-10-8-15-11-18(21)19(22)12-16(15)17(13-20)14-6-4-3-5-7-14/h2-7,11-12,17,21-22H,1,8-10,13H2 | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2.11E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SRI International
Curated by PDSP Ki Database
| |
NIDA Res Monogr 178: 440-66 (1998)
BindingDB Entry DOI: 10.7270/Q23J3BH2 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Rattus norvegicus (rat)) | BDBM50004918

((+/-)-APD3-Allyl-1-phenyl-2,3,4,5-tetrahydro-1H-be...)Show InChI InChI=1S/C19H21NO2/c1-2-9-20-10-8-15-11-18(21)19(22)12-16(15)17(13-20)14-6-4-3-5-7-14/h2-7,11-12,17,21-22H,1,8-10,13H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SRI International
Curated by PDSP Ki Database
| |
NIDA Res Monogr 178: 440-66 (1998)
BindingDB Entry DOI: 10.7270/Q23J3BH2 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Rattus norvegicus (rat)) | BDBM50004918

((+/-)-APD3-Allyl-1-phenyl-2,3,4,5-tetrahydro-1H-be...)Show InChI InChI=1S/C19H21NO2/c1-2-9-20-10-8-15-11-18(21)19(22)12-16(15)17(13-20)14-6-4-3-5-7-14/h2-7,11-12,17,21-22H,1,8-10,13H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard University
Curated by PDSP Ki Database
| |
Eur J Pharmacol 474: 137-40 (2003)
Article DOI: 10.1016/s0014-2999(03)02008-9
BindingDB Entry DOI: 10.7270/Q2DN43MQ |
More data for this
Ligand-Target Pair | |
Serotonin receptor 2a and 2c (5HT2A and 5HT2C)
(Rattus norvegicus (Rat)) | BDBM50004918

((+/-)-APD3-Allyl-1-phenyl-2,3,4,5-tetrahydro-1H-be...)Show InChI InChI=1S/C19H21NO2/c1-2-9-20-10-8-15-11-18(21)19(22)12-16(15)17(13-20)14-6-4-3-5-7-14/h2-7,11-12,17,21-22H,1,8-10,13H2 | KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 3.04E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SRI International
Curated by PDSP Ki Database
| |
NIDA Res Monogr 178: 440-66 (1998)
BindingDB Entry DOI: 10.7270/Q23J3BH2 |
More data for this
Ligand-Target Pair | |
D(1A) dopamine receptor
(Homo sapiens (Human)) | BDBM50004918

((+/-)-APD3-Allyl-1-phenyl-2,3,4,5-tetrahydro-1H-be...)Show InChI InChI=1S/C19H21NO2/c1-2-9-20-10-8-15-11-18(21)19(22)12-16(15)17(13-20)14-6-4-3-5-7-14/h2-7,11-12,17,21-22H,1,8-10,13H2 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a |
ShanghaiTech University
Curated by ChEMBL
| Assay Description
Agonist activity at human dopamine D1 receptor expressed in CHOK1 cells assessed as assessed as increase in beta-arrestin-2 recruitment after 60 mins... |
J Med Chem 61: 9841-9878 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00435 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50004918

((+/-)-APD3-Allyl-1-phenyl-2,3,4,5-tetrahydro-1H-be...)Show InChI InChI=1S/C19H21NO2/c1-2-9-20-10-8-15-11-18(21)19(22)12-16(15)17(13-20)14-6-4-3-5-7-14/h2-7,11-12,17,21-22H,1,8-10,13H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Dopamine receptor D2 agonist activity was determined for inhibition of the constrictor response to electrical stimulation in an isolated perfused rab... |
J Med Chem 30: 35-40 (1987)
BindingDB Entry DOI: 10.7270/Q2X92BV6 |
More data for this
Ligand-Target Pair | |
D(1A) dopamine receptor
(RAT) | BDBM50004918

((+/-)-APD3-Allyl-1-phenyl-2,3,4,5-tetrahydro-1H-be...)Show InChI InChI=1S/C19H21NO2/c1-2-9-20-10-8-15-11-18(21)19(22)12-16(15)17(13-20)14-6-4-3-5-7-14/h2-7,11-12,17,21-22H,1,8-10,13H2 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 25 | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Dopamine agonist (Dopamine receptor D1) activity was measured as increase in cAMP formation relative to maximum increase in dopamine-sensitive adenyl... |
J Med Chem 30: 35-40 (1987)
BindingDB Entry DOI: 10.7270/Q2X92BV6 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Rattus norvegicus (rat)) | BDBM50004918

((+/-)-APD3-Allyl-1-phenyl-2,3,4,5-tetrahydro-1H-be...)Show InChI InChI=1S/C19H21NO2/c1-2-9-20-10-8-15-11-18(21)19(22)12-16(15)17(13-20)14-6-4-3-5-7-14/h2-7,11-12,17,21-22H,1,8-10,13H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 2.43E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Research Biochemicals Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM-09151-2 (60 pm) from dopamine receptor D2 in crude membrane fraction of rat brain corpus striatum |
J Med Chem 35: 1466-71 (1992)
BindingDB Entry DOI: 10.7270/Q26H4GCW |
More data for this
Ligand-Target Pair | |
D(1A) dopamine receptor
(Homo sapiens (Human)) | BDBM50004918

((+/-)-APD3-Allyl-1-phenyl-2,3,4,5-tetrahydro-1H-be...)Show InChI InChI=1S/C19H21NO2/c1-2-9-20-10-8-15-11-18(21)19(22)12-16(15)17(13-20)14-6-4-3-5-7-14/h2-7,11-12,17,21-22H,1,8-10,13H2 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a |
ShanghaiTech University
Curated by ChEMBL
| Assay Description
Agonist activity at human dopamine D1 receptor expressed in CHOK1 cells assessed as reversal of Ro 20-1724 mediated decrease in cAMP accumulation aft... |
J Med Chem 61: 9841-9878 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00435 |
More data for this
Ligand-Target Pair | |
D(1A) dopamine receptor
(RAT) | BDBM50004918

((+/-)-APD3-Allyl-1-phenyl-2,3,4,5-tetrahydro-1H-be...)Show InChI InChI=1S/C19H21NO2/c1-2-9-20-10-8-15-11-18(21)19(22)12-16(15)17(13-20)14-6-4-3-5-7-14/h2-7,11-12,17,21-22H,1,8-10,13H2 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Research Biochemicals Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-SCH- 23390 (0.3 nM) from dopamine receptor D1 in crude membrane fraction of rat brain corpus striatum |
J Med Chem 35: 1466-71 (1992)
BindingDB Entry DOI: 10.7270/Q26H4GCW |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data