 Found 5 hits for monomerid = 50008760
Found 5 hits for monomerid = 50008760 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50008760
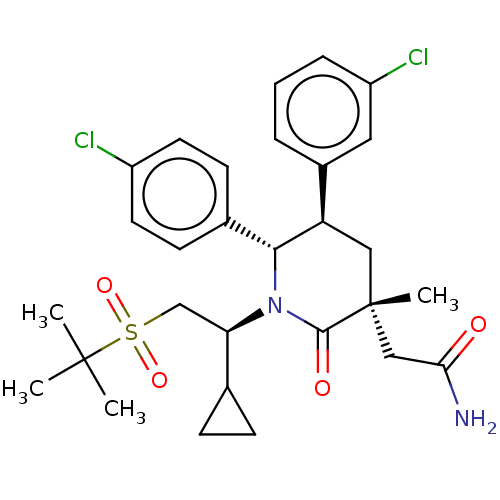
(CHEMBL3236364)Show SMILES CC(C)(C)S(=O)(=O)C[C@H](C1CC1)N1[C@@H]([C@H](C[C@](C)(CC(N)=O)C1=O)c1cccc(Cl)c1)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C29H36Cl2N2O4S/c1-28(2,3)38(36,37)17-24(18-8-9-18)33-26(19-10-12-21(30)13-11-19)23(20-6-5-7-22(31)14-20)15-29(4,27(33)35)16-25(32)34/h5-7,10-14,18,23-24,26H,8-9,15-17H2,1-4H3,(H2,32,34)/t23-,24-,26-,29-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| US Patent
| n/a | n/a | 1.83 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Amgen Inc.
US Patent
| Assay Description
The standard assay conditions for the in vitro HTRF assay consisted of a 50 ul total reaction volume in black 384-well Costar polypropylene plates in... |
US Patent US8952036 (2015)
BindingDB Entry DOI: 10.7270/Q27P8X4P |
More data for this
Ligand-Target Pair | |
E3 ubiquitin-protein ligase Mdm2
(Homo sapiens (Human)) | BDBM50008760

(CHEMBL3236364)Show SMILES CC(C)(C)S(=O)(=O)C[C@H](C1CC1)N1[C@@H]([C@H](C[C@](C)(CC(N)=O)C1=O)c1cccc(Cl)c1)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C29H36Cl2N2O4S/c1-28(2,3)38(36,37)17-24(18-8-9-18)33-26(19-10-12-21(30)13-11-19)23(20-6-5-7-22(31)14-20)15-29(4,27(33)35)16-25(32)34/h5-7,10-14,18,23-24,26H,8-9,15-17H2,1-4H3,(H2,32,34)/t23-,24-,26-,29-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to GST-thrombin-tagged human MDM2 (1 to 188) expressed in Escherichia coli assessed as inhibition of interaction with p53 in serum f... |
J Med Chem 57: 2963-88 (2014)
Article DOI: 10.1021/jm401911v
BindingDB Entry DOI: 10.7270/Q2862J0J |
More data for this
Ligand-Target Pair | |
E3 ubiquitin-protein ligase Mdm2
(Homo sapiens (Human)) | BDBM50008760

(CHEMBL3236364)Show SMILES CC(C)(C)S(=O)(=O)C[C@H](C1CC1)N1[C@@H]([C@H](C[C@](C)(CC(N)=O)C1=O)c1cccc(Cl)c1)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C29H36Cl2N2O4S/c1-28(2,3)38(36,37)17-24(18-8-9-18)33-26(19-10-12-21(30)13-11-19)23(20-6-5-7-22(31)14-20)15-29(4,27(33)35)16-25(32)34/h5-7,10-14,18,23-24,26H,8-9,15-17H2,1-4H3,(H2,32,34)/t23-,24-,26-,29-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.170 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to human GST-thrombin-tagged MDM2 ( 1 to 188 aa) assessed as inhibition of interaction with human p53 preincubated with compound for... |
J Med Chem 57: 10499-511 (2014)
Article DOI: 10.1021/jm501550p
BindingDB Entry DOI: 10.7270/Q2571DPW |
More data for this
Ligand-Target Pair | |
E3 ubiquitin-protein ligase Mdm2
(Homo sapiens (Human)) | BDBM50008760

(CHEMBL3236364)Show SMILES CC(C)(C)S(=O)(=O)C[C@H](C1CC1)N1[C@@H]([C@H](C[C@](C)(CC(N)=O)C1=O)c1cccc(Cl)c1)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C29H36Cl2N2O4S/c1-28(2,3)38(36,37)17-24(18-8-9-18)33-26(19-10-12-21(30)13-11-19)23(20-6-5-7-22(31)14-20)15-29(4,27(33)35)16-25(32)34/h5-7,10-14,18,23-24,26H,8-9,15-17H2,1-4H3,(H2,32,34)/t23-,24-,26-,29-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to GST-thrombin-tagged human MDM2 (1 to 188) expressed in Escherichia coli assessed as inhibition of interaction with p53 in buffer ... |
J Med Chem 57: 2963-88 (2014)
Article DOI: 10.1021/jm401911v
BindingDB Entry DOI: 10.7270/Q2862J0J |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50008760

(CHEMBL3236364)Show SMILES CC(C)(C)S(=O)(=O)C[C@H](C1CC1)N1[C@@H]([C@H](C[C@](C)(CC(N)=O)C1=O)c1cccc(Cl)c1)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C29H36Cl2N2O4S/c1-28(2,3)38(36,37)17-24(18-8-9-18)33-26(19-10-12-21(30)13-11-19)23(20-6-5-7-22(31)14-20)15-29(4,27(33)35)16-25(32)34/h5-7,10-14,18,23-24,26H,8-9,15-17H2,1-4H3,(H2,32,34)/t23-,24-,26-,29-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) using midazolam as substrate |
J Med Chem 57: 2963-88 (2014)
Article DOI: 10.1021/jm401911v
BindingDB Entry DOI: 10.7270/Q2862J0J |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data