

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50012434 (REV-5,901)1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hexan-1-ol::1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hexan-1-ol::1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hexan-1-ol (RG 5901)::1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hexan-1-ol(REV-5901)::CHEMBL8747::REV-5901::REV-901
SMILES: CCCCCC(O)c1cccc(OCc2ccc3ccccc3n2)c1
InChI Key: InChIKey=JRLOEMCOOZSCQP-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cysteinyl leukotriene receptor 1 (GUINEA PIG) | BDBM50012434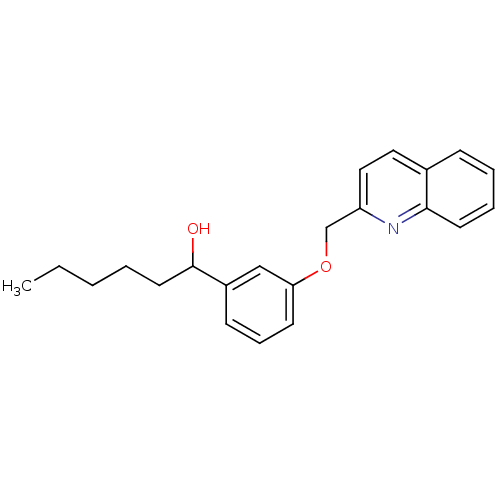 ((REV-5,901)1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hex...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rorer Central Research Curated by ChEMBL | Assay Description Binding affinity against Cysteinyl leukotriene D4 receptor from guinea pig lung was determined using [3H]-LTD4 (0.2 nM) | J Med Chem 33: 1186-94 (1990) BindingDB Entry DOI: 10.7270/Q2TB15WM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cysteinyl leukotriene receptor 1 (GUINEA PIG) | BDBM50012434 ((REV-5,901)1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hex...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article | 8.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Compound was tested for the inhibition of [3H]-leukotriene D4 binding to LTD4 receptor in guinea pig lung membranes | Bioorg Med Chem Lett 5: 1371-1376 (1995) Article DOI: 10.1016/0960-894X(95)00226-J BindingDB Entry DOI: 10.7270/Q2QR4X29 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cysteinyl leukotriene receptor 1 (GUINEA PIG) | BDBM50012434 ((REV-5,901)1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hex...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article | 8.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of [3H]-LTD4 binding to LTD4 receptor in guinea pig lung membranes | Bioorg Med Chem Lett 5: 1365-1370 (1995) Article DOI: 10.1016/0960-894X(95)00225-I BindingDB Entry DOI: 10.7270/Q2VH5NSM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Arachidonate 5-lipoxygenase (Homo sapiens (Human)) | BDBM50012434 ((REV-5,901)1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hex...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Laboratories Curated by ChEMBL | Assay Description Ability to inhibit 5-lipoxygenase in guinea pig | J Med Chem 30: 2087-93 (1987) BindingDB Entry DOI: 10.7270/Q2FQ9VMQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Arachidonate 5-lipoxygenase (Rattus norvegicus) | BDBM50012434 ((REV-5,901)1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hex...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Ability to inhibit Lipoxygenase in vitro was determined | J Med Chem 30: 400-5 (1987) BindingDB Entry DOI: 10.7270/Q2K936JS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Arachidonate 5-lipoxygenase (Homo sapiens (Human)) | BDBM50012434 ((REV-5,901)1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hex...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | n/a | n/a | >100 | n/a | n/a | n/a | n/a |
ICI Pharma Curated by ChEMBL | Assay Description Ex vivo inhibition of LTB4 production was measured in dog blood | J Med Chem 34: 1028-36 (1991) BindingDB Entry DOI: 10.7270/Q28G8MXH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Arachidonate 5-lipoxygenase (Rattus norvegicus) | BDBM50012434 ((REV-5,901)1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hex...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article | n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of 5-Lipoxygenase (5-LO) in rat basophilic leukemic cells | Bioorg Med Chem Lett 5: 1365-1370 (1995) Article DOI: 10.1016/0960-894X(95)00225-I BindingDB Entry DOI: 10.7270/Q2VH5NSM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Arachidonate 5-lipoxygenase (Rattus norvegicus) | BDBM50012434 ((REV-5,901)1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hex...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | >4.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Compound was tested for its in vitro inhibitory activity against RBL-1 5-LO (insuluble above 45 uM) | J Med Chem 30: 574-80 (1987) BindingDB Entry DOI: 10.7270/Q2QN67BB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Arachidonate 5-lipoxygenase (Rattus norvegicus) | BDBM50012434 ((REV-5,901)1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hex...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | >4.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description In vitro inhibitory activity against 5-lipoxygenase from rat basophilic leukemia cells. | J Med Chem 30: 1177-86 (1987) BindingDB Entry DOI: 10.7270/Q25M668Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Arachidonate 5-lipoxygenase (Rattus norvegicus) | BDBM50012434 ((REV-5,901)1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hex...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 160 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research Curated by ChEMBL | Assay Description The compound was tested for inhibitory activity against 5-lipoxygenase in rat polymorphonuclear leukocytes[PMNS] (in vivo) | J Med Chem 35: 2501-24 (1992) BindingDB Entry DOI: 10.7270/Q23B60R4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cyclooxygenase (Homo sapiens (Human)) | BDBM50012434 ((REV-5,901)1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hex...) | PDB KEGG UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 5.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Laboratories Curated by ChEMBL | Assay Description Ability to inhibit Prostaglandin G/H synthase in guinea pig | J Med Chem 30: 2087-93 (1987) BindingDB Entry DOI: 10.7270/Q2FQ9VMQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Arachidonate 5-lipoxygenase (Rattus norvegicus) | BDBM50012434 ((REV-5,901)1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hex...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
ICI Pharma Curated by ChEMBL | Assay Description In vitro inhibition of LTB4 production was measured in rat blood | J Med Chem 34: 1028-36 (1991) BindingDB Entry DOI: 10.7270/Q28G8MXH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Arachidonate 5-lipoxygenase (Rattus norvegicus) | BDBM50012434 ((REV-5,901)1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hex...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 120 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat neutrophil 5- Lipoxygenase (5-LO) | J Med Chem 30: 96-104 (1987) BindingDB Entry DOI: 10.7270/Q2PV6MKS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cyclooxygenase (RAT) | BDBM50012434 ((REV-5,901)1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hex...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | >3.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
ICI Pharma Curated by ChEMBL | Assay Description In vitro inhibition of PGE-2 production was measured in rat blood | J Med Chem 34: 1028-36 (1991) BindingDB Entry DOI: 10.7270/Q28G8MXH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Arachidonate 5-lipoxygenase (Rattus norvegicus) | BDBM50012434 ((REV-5,901)1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hex...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat polymorphonuclear leukocyte (PMN) 5-Lipoxygenase in vitro | J Med Chem 29: 1429-35 (1986) BindingDB Entry DOI: 10.7270/Q2GX49K4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Arachidonate 5-lipoxygenase (Homo sapiens (Human)) | BDBM50012434 ((REV-5,901)1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hex...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Rorer Central Research Curated by ChEMBL | Assay Description Inhibition against 5-lipoxygenase from guinea pig polymorphonuclear lymphocytes | J Med Chem 33: 1186-94 (1990) BindingDB Entry DOI: 10.7270/Q2TB15WM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Arachidonate 5-lipoxygenase (Rattus norvegicus) | BDBM50012434 ((REV-5,901)1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hex...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | >4.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description In vitro inhibition of 5-lipoxygenase (5-lo) from the 20000 g supernatant of RBI-1 cells | J Med Chem 34: 2158-65 (1991) BindingDB Entry DOI: 10.7270/Q2NS0SWM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cysteinyl leukotriene receptor (Homo sapiens (Human)) | BDBM50012434 ((REV-5,901)1-[3-(Quinolin-2-ylmethoxy)-phenyl]-hex...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 510 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase II | J Med Chem 35: 3832-44 (1992) BindingDB Entry DOI: 10.7270/Q2TQ625N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||