

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50017681 (1S,2R,4S,5R)-2-[(S)-Hydroxy-(6-methoxy-quinolin-4-yl)-methyl]-5-vinyl-1-azonia-bicyclo[2.2.2]octane::(6-Methoxy-quinolin-4-yl)-((2S,4S,5R)-5-vinyl-1-aza-bicyclo[2.2.2]oct-2-yl)-methanol::(6-Methoxy-quinolin-4-yl)-(5-vinyl-1-aza-bicyclo[2.2.2]oct-2-yl)-methanol::(6-Methoxy-quinolin-4-yl)-(5-vinyl-1-aza-bicyclo[2.2.2]oct-2-yl)-methanol; compound with sulfuric acid::(6-Methoxy-quinolin-4-yl)-(5-vinyl-1-aza-bicyclo[2.2.2]oct-2-yl)-methanol; sulphate::(S)-(6-Methoxy-quinolin-4-yl)-((2R,4S,5R)-5-vinyl-1-aza-bicyclo[2.2.2]oct-2-yl)-methanol::(S)-(6-Methoxy-quinolin-4-yl)-((2R,4S,5S)-5-vinyl-1-aza-bicyclo[2.2.2]oct-2-yl)-methanol::(S)-(6-Methoxy-quinolin-4-yl)-((2R,5R)-5-vinyl-1-aza-bicyclo[2.2.2]oct-2-yl)-methanol::(S)-(6-Methoxy-quinolin-4-yl)-(5-vinyl-1-aza-bicyclo[2.2.2]oct-2-yl)-methanol::(S)-(6-methoxy-quinolin-4-yl)-((2R,5S)-5-vinyl-1-aza-bicyclo[2.2.2]oct-2-yl)-methanol::(S)-(6-methoxyquinolin-4-yl)((2R,4S,5R)-5-vinylquinuclidin-2-yl)methanol::(S)-(6-methoxyquinolin-4-yl)((2R,5R)-5-vinylquinuclidin-2-yl)methanol::(S)-(6-methoxyquinolin-4-yl)((3S,4R,7R)-3-vinylquinuclidin-7-yl)methanol::(S)-(6-methoxyquinolin-4-yl)[(2R,4S,5R)-5-vinyl-1-azabicyclo[2.2.2]oct-2-yl]methanol::(quinidine)(6-Methoxy-quinolin-4-yl)-(5-vinyl-1-aza-bicyclo[2.2.2]oct-2-yl)-methanol::6-methoxy-4-quinolyl-5-vinyl-1-azabicyclo[2.2.2]oct-2-yl-S-methanol::8R,9S(6-Methoxy-quinolin-4-yl)-(5-vinyl-1-aza-bicyclo[2.2.2]oct-2-yl)-methanol::CHEMBL97::CIN-QUIN::Cardioquin::Duraquin::QUINIDINE::Quinact::Quinaglute::Quinalan::Quinatime::Quinidex::Quinine::Quinora::TCMDC-131239
SMILES: COc1ccc2nccc([C@H](O)C3CC4CCN3CC4C=C)c2c1
InChI Key: InChIKey=LOUPRKONTZGTKE-LHFMAAMQSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cytochrome P450 2D6 (Homo sapiens (Human)) | BDBM50017681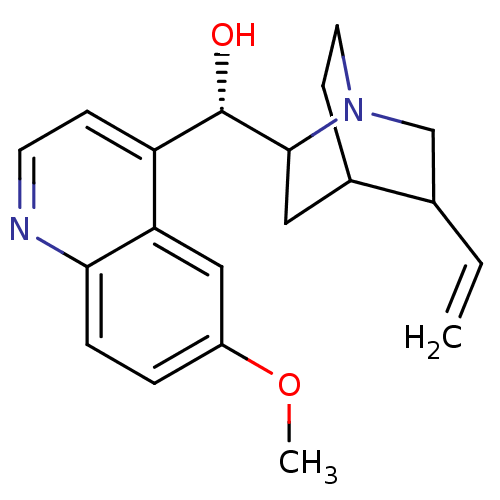 ((1S,2R,4S,5R)-2-[(S)-Hydroxy-(6-methoxy-quinolin-4...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institut f£r Toxikologie Curated by ChEMBL | Assay Description Inhibition of 1'-hydroxybufuralol formation by human liver microsomes | J Med Chem 36: 1136-45 (1993) BindingDB Entry DOI: 10.7270/Q2GM87X6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D6 (Homo sapiens (Human)) | BDBM50017681 ((1S,2R,4S,5R)-2-[(S)-Hydroxy-(6-methoxy-quinolin-4...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 43 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institut f£r Toxikologie Curated by ChEMBL | Assay Description Inhibition of partially purified cytochrome P450 2D6 1'-hydroxybufuralol formation | J Med Chem 36: 1136-45 (1993) BindingDB Entry DOI: 10.7270/Q2GM87X6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D6 (Homo sapiens (Human)) | BDBM50017681 ((1S,2R,4S,5R)-2-[(S)-Hydroxy-(6-methoxy-quinolin-4...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 43 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institut f£r Toxikologie Curated by ChEMBL | Assay Description Inhibition of partially purified cytochrome P450 2D6 1'-hydroxybufuralol formation | J Med Chem 36: 1136-45 (1993) BindingDB Entry DOI: 10.7270/Q2GM87X6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D6 (Homo sapiens (Human)) | BDBM50017681 ((1S,2R,4S,5R)-2-[(S)-Hydroxy-(6-methoxy-quinolin-4...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institut f£r Toxikologie Curated by ChEMBL | Assay Description Inhibitory effect on Bufuralol 1'-hydroxylation by human liver microsomes (Ki = apparent inhibition constant) | J Med Chem 36: 1136-45 (1993) BindingDB Entry DOI: 10.7270/Q2GM87X6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adrenaline alpha1 (RAT) | BDBM50017681 ((1S,2R,4S,5R)-2-[(S)-Hydroxy-(6-methoxy-quinolin-4...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 6.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Heidelberg Curated by ChEMBL | Assay Description TP_TRANSPORTER: inhibition of MPP+ uptake in OCT1-expressing HEK293 cells | Naunyn Schmiedebergs Arch Pharmacol 354: 320-6 (1997) Article DOI: 10.1007/bf00171063 BindingDB Entry DOI: 10.7270/Q2MG7QSP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-glycoprotein 1 (Homo sapiens (Human)) | BDBM50017681 ((1S,2R,4S,5R)-2-[(S)-Hydroxy-(6-methoxy-quinolin-4...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 8.59E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Kansas Curated by ChEMBL | Assay Description TP_TRANSPORTER: inhibition of Digoxin transepithelial transport (basal to apical) in MDR1-expressing MDCK cells | Pharm Res 19: 765-72 (2002) BindingDB Entry DOI: 10.7270/Q2BZ67B1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adrenaline alpha1 (RAT) | BDBM50017681 ((1S,2R,4S,5R)-2-[(S)-Hydroxy-(6-methoxy-quinolin-4...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 1.46E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University Hospital Curated by ChEMBL | Assay Description TP_TRANSPORTER: inhibition of TEA uptake (basolateral to cell) in OCT1-expressing MDCK cells | J Pharmacol Exp Ther 287: 800-5 (1998) BindingDB Entry DOI: 10.7270/Q2D79CQ4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adrenaline alpha2 (RAT) | BDBM50017681 ((1S,2R,4S,5R)-2-[(S)-Hydroxy-(6-methoxy-quinolin-4...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 1.91E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University Hospital Curated by ChEMBL | Assay Description TP_TRANSPORTER: inhibition of TEA uptake (basolateral to cell) in OCT2-expressing MDCK cells | J Pharmacol Exp Ther 287: 800-5 (1998) BindingDB Entry DOI: 10.7270/Q2D79CQ4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2C9 (Homo sapiens (Human)) | BDBM50017681 ((1S,2R,4S,5R)-2-[(S)-Hydroxy-(6-methoxy-quinolin-4...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 3.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D Curated by ChEMBL | Assay Description Binding affinity towards cytochrome P450 2C9 | J Med Chem 47: 907-14 (2004) Article DOI: 10.1021/jm030972s BindingDB Entry DOI: 10.7270/Q2ZK5HF3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adrenaline alpha1 (RAT) | BDBM50017681 ((1S,2R,4S,5R)-2-[(S)-Hydroxy-(6-methoxy-quinolin-4...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.27E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Anatomy of the Bayerische Julius-Maximilians-Universit£t Curated by ChEMBL | Assay Description TP_TRANSPORTER: inhibition of TEA uptake (TEA: 10 uM) in Xenopus laevis oocytes | Am J Physiol Renal Physiol 281: 454-68 (2001) Article DOI: 10.1152/ajprenal.2001.281.3.F454 BindingDB Entry DOI: 10.7270/Q2BR8TF3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-glycoprotein 1 (Homo sapiens (Human)) | BDBM50017681 ((1S,2R,4S,5R)-2-[(S)-Hydroxy-(6-methoxy-quinolin-4...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute Curated by ChEMBL | Assay Description TP_TRANSPORTER: inhibition of LDS-751 efflux in NIH-3T3-G185 cells | Biochem Biophys Res Commun 289: 580-5 (2001) Article DOI: 10.1006/bbrc.2001.6000 BindingDB Entry DOI: 10.7270/Q21N82C8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-glycoprotein 1 (Homo sapiens (Human)) | BDBM50017681 ((1S,2R,4S,5R)-2-[(S)-Hydroxy-(6-methoxy-quinolin-4...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.88E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute Curated by ChEMBL | Assay Description TP_TRANSPORTER: inhibition of Daunorubicin efflux in NIH-3T3-G185 cells | Biochem Biophys Res Commun 289: 580-5 (2001) Article DOI: 10.1006/bbrc.2001.6000 BindingDB Entry DOI: 10.7270/Q21N82C8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-glycoprotein 1 (Homo sapiens (Human)) | BDBM50017681 ((1S,2R,4S,5R)-2-[(S)-Hydroxy-(6-methoxy-quinolin-4...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3.39E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute Curated by ChEMBL | Assay Description TP_TRANSPORTER: inhibition of Rhodamine 123 efflux in NIH-3T3-G185 cells | Biochem Biophys Res Commun 289: 580-5 (2001) Article DOI: 10.1006/bbrc.2001.6000 BindingDB Entry DOI: 10.7270/Q21N82C8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adrenaline alpha2 (RAT) | BDBM50017681 ((1S,2R,4S,5R)-2-[(S)-Hydroxy-(6-methoxy-quinolin-4...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.46E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Anatomy of the Bayerische Julius-Maximilians-Universit£t Curated by ChEMBL | Assay Description TP_TRANSPORTER: inhibition of TEA uptake (TEA: 10 uM) in Xenopus laevis oocytes | Am J Physiol Renal Physiol 281: 454-68 (2001) Article DOI: 10.1152/ajprenal.2001.281.3.F454 BindingDB Entry DOI: 10.7270/Q2BR8TF3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-glycoprotein 1 (Homo sapiens (Human)) | BDBM50017681 ((1S,2R,4S,5R)-2-[(S)-Hydroxy-(6-methoxy-quinolin-4...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 2.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt University School of Medicine Curated by ChEMBL | Assay Description TP_TRANSPORTER: inhibition of Digoxin transepithelial transport (basal to apical) (Digoxin: 5 uM) in Caco-2 cells | Drug Metab Dispos 28: 655-60 (2000) BindingDB Entry DOI: 10.7270/Q2959JTX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||