

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50023364 CHEMBL3360785
SMILES: [H][C@@]1([#6]-[#6][C@@]2([H])\[#6](-[#6]-[#6]-[#6][C@]12[#6])=[#6]\[#6]=[#6]-1\[#6]-[#6@@H](-[#8])-[#6](=[#6])-[#6@H](-[#8])-[#6]-1)[#6@H](-[#6])-[#6]-[#6]-[#6]-[#7]-[#6]-1-[#6]-[#6]-1
InChI Key: InChIKey=FLEHQRTTWKDNGI-WSGFCWAJSA-N
Data: 1 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cytochrome P450 24A1 (Homo sapiens (Human)) | BDBM50023364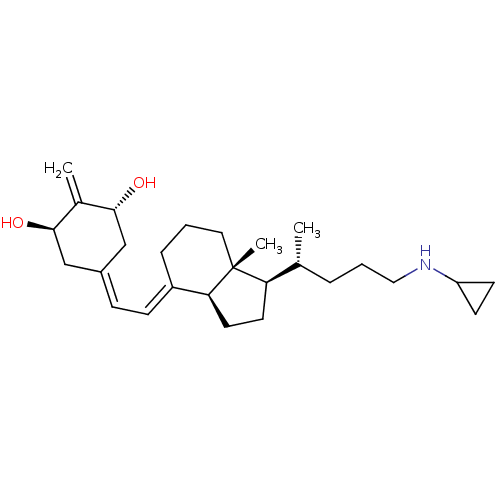 (CHEMBL3360785) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 42 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cardiff University Curated by ChEMBL | Assay Description Inhibition of human MBP-tagged CYP24A1 expressed in Escherichia coli using 1,25(OH)2D3 substrate in presence of bovine adrenodoxin, adrenodoxin reduc... | J Med Chem 57: 7702-15 (2014) Article DOI: 10.1021/jm5009314 BindingDB Entry DOI: 10.7270/Q2DN46ND | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||