

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50025356 4a,6a-Dimethyl-2-oxo-2,4a,4b,5,6,6a,7,8,9,9a,9b,10,11,11a-tetradecahydro-1H-indeno[5,4-f]quinoline-7-carboxylic acid tert-butylamide::CHEMBL710::Finasteride::N-tert-butyl-2,15-dimethyl-5-oxo-6-azatetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-3-ene-14-carboxamide
SMILES: CC(C)(C)NC(=O)C1CCC2C3CCC4NC(=O)C=CC4(C)C3CCC12C
InChI Key: InChIKey=DBEPLOCGEIEOCV-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-alpha-Reductase (5α-Reductase) (Homo sapiens (Human)) | BDBM50025356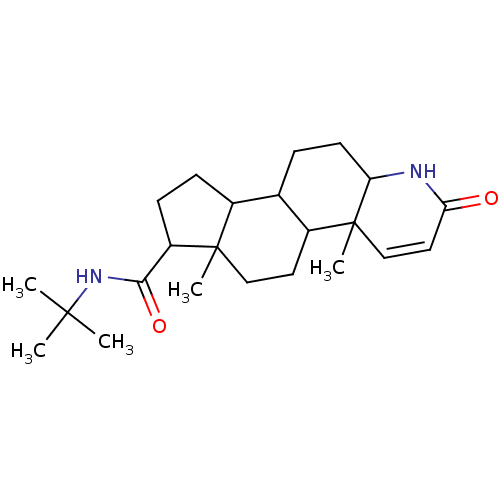 (4a,6a-Dimethyl-2-oxo-2,4a,4b,5,6,6a,7,8,9,9a,9b,10...) | UniProtKB/TrEMBL GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | 6.5 | 37 |
Metropolitan University-Xochimilco | Assay Description The reaction mixtures contained a final volume of 1 mL, 1 mM DTT, sodium phosphate buffer 40 mM, at pH 6.5, 2 mM, NADPH, 2 nM [1,2,6,7-3H]T or [1,2,3... | J Enzyme Inhib Med Chem 26: 712-9 (2011) Article DOI: 10.3109/14756366.2010.548330 BindingDB Entry DOI: 10.7270/Q25719XS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Steroid 5-alpha-reductase (Homo sapiens (Human)) | BDBM50025356 (4a,6a-Dimethyl-2-oxo-2,4a,4b,5,6,6a,7,8,9,9a,9b,10...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Inc. Research Institute Curated by ChEMBL | Assay Description Inhibition of recombinant human 5-alpha reductase-2 at a concentration of 5 microL after preincubation for 10 minutes | J Med Chem 36: 4313-5 (1994) BindingDB Entry DOI: 10.7270/Q2PK0F76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5α-Reductase 1 (5α-R1) (Homo sapiens (Human)) | BDBM50025356 (4a,6a-Dimethyl-2-oxo-2,4a,4b,5,6,6a,7,8,9,9a,9b,10...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Inc. Research Institute Curated by ChEMBL | Assay Description Inhibition of recombinant human 5-alpha reductase-1 at a concentration of 5 microL after preincubation for 10 minutes | J Med Chem 36: 4313-5 (1994) BindingDB Entry DOI: 10.7270/Q2PK0F76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Steroid 5-alpha-reductase (Homo sapiens (Human)) | BDBM50025356 (4a,6a-Dimethyl-2-oxo-2,4a,4b,5,6,6a,7,8,9,9a,9b,10...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Florence Curated by ChEMBL | Assay Description Inhibitory concentration against human recombinant steroid 5-alpha-reductase type 2 in stably transfected chinese hamster ovary 1827 cells using [3H]... | J Med Chem 47: 3546-60 (2004) Article DOI: 10.1021/jm031131o BindingDB Entry DOI: 10.7270/Q24J0DKZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Steroid 5-alpha-reductase (Rattus norvegicus) | BDBM50025356 (4a,6a-Dimethyl-2-oxo-2,4a,4b,5,6,6a,7,8,9,9a,9b,10...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article | n/a | n/a | 4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Compound was tested for its inhibitory potency against rat Steroid 5-alpha-reductase type 1 expressed in transformed yeast Saccharomyces cerevesiae | Bioorg Med Chem Lett 6: 1997-2002 (1996) Article DOI: 10.1016/0960-894X(96)00360-5 BindingDB Entry DOI: 10.7270/Q2K0748S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Steroid 5-alpha-reductase (Homo sapiens (Human)) | BDBM50025356 (4a,6a-Dimethyl-2-oxo-2,4a,4b,5,6,6a,7,8,9,9a,9b,10...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Canterbury Curated by ChEMBL | Assay Description Inhibition of steroid 5alpha reductase type 2 expressed in HEK 293 cells | Bioorg Med Chem Lett 17: 3603-7 (2007) Article DOI: 10.1016/j.bmcl.2007.04.049 BindingDB Entry DOI: 10.7270/Q2P84BKV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5α-Reductase 1 (5α-R1) (Homo sapiens (Human)) | BDBM50025356 (4a,6a-Dimethyl-2-oxo-2,4a,4b,5,6,6a,7,8,9,9a,9b,10...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 453 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Canterbury Curated by ChEMBL | Assay Description Inhibition of steroid 5alpha reductase type 1 expressed in HEK 293 cells | Bioorg Med Chem Lett 17: 3603-7 (2007) Article DOI: 10.1016/j.bmcl.2007.04.049 BindingDB Entry DOI: 10.7270/Q2P84BKV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Steroid 5-alpha-reductase (Homo sapiens (Human)) | BDBM50025356 (4a,6a-Dimethyl-2-oxo-2,4a,4b,5,6,6a,7,8,9,9a,9b,10...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article | n/a | n/a | 4.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory activity of the compound against type-2 human steroid 5-alpha-reductase. | Bioorg Med Chem Lett 6: 481-484 (1996) Article DOI: 10.1016/0960-894X(96)00054-6 BindingDB Entry DOI: 10.7270/Q2BZ661K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5α-Reductase 1 (5α-R1) (Homo sapiens (Human)) | BDBM50025356 (4a,6a-Dimethyl-2-oxo-2,4a,4b,5,6,6a,7,8,9,9a,9b,10...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article | n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory activity of the compound against type-1 human steroid 5-alpha-reductase. | Bioorg Med Chem Lett 6: 481-484 (1996) Article DOI: 10.1016/0960-894X(96)00054-6 BindingDB Entry DOI: 10.7270/Q2BZ661K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5α-Reductase 1 (5α-R1) (Homo sapiens (Human)) | BDBM50025356 (4a,6a-Dimethyl-2-oxo-2,4a,4b,5,6,6a,7,8,9,9a,9b,10...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 911 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Florence Curated by ChEMBL | Assay Description Inhibitory concentration against human recombinant steroid 5-alpha-reductase type I in stably transfected chinese hamster ovary (CHO) 1827 cells usin... | J Med Chem 47: 3546-60 (2004) Article DOI: 10.1021/jm031131o BindingDB Entry DOI: 10.7270/Q24J0DKZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||