

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50034641 CHEMBL3360415
SMILES: COc1ccc(NC(=O)[C@@H](C)Nc2nc3n(ncc3c(=O)[nH]2)C2CCCC2)cc1
InChI Key: InChIKey=UBBCZBKJYNXHGT-GFCCVEGCSA-N
Data: 9 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Phosphodiesterase 9A (Homo sapiens (Human)) | BDBM50034641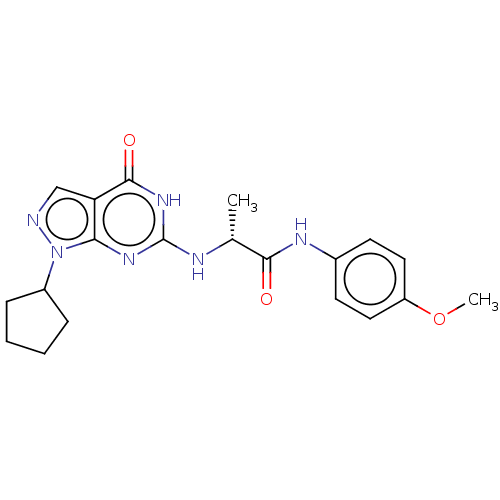 (CHEMBL3360415) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
Sun Yat-Sen University Curated by ChEMBL | Assay Description Inhibition of PDE9A2 catalytic domain (unknown origin) using [3H]-cGMP/[3H]-cAMP as substrate after 15 mins by liquid scintillation counting analysis | J Med Chem 57: 10304-13 (2014) Article DOI: 10.1021/jm500836h BindingDB Entry DOI: 10.7270/Q28P624W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Calcium/calmodulin-dependent 3',5'-cyclic nucleotide phosphodiesterase 1B (Homo sapiens (Human)) | BDBM50034641 (CHEMBL3360415) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 473 | n/a | n/a | n/a | n/a | n/a | n/a |
Sun Yat-Sen University Curated by ChEMBL | Assay Description Inhibition of PDE1B2 catalytic domain (unknown origin) using [3H]-cGMP/[3H]-cAMP as substrate after 15 mins by liquid scintillation counting analysis | J Med Chem 57: 10304-13 (2014) Article DOI: 10.1021/jm500836h BindingDB Entry DOI: 10.7270/Q28P624W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cGMP-specific 3',5'-cyclic phosphodiesterase (Homo sapiens (Human)) | BDBM50034641 (CHEMBL3360415) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 91 | n/a | n/a | n/a | n/a | n/a | n/a |
Sun Yat-Sen University Curated by ChEMBL | Assay Description Inhibition of PDE5A1 catalytic domain (unknown origin) using [3H]-cGMP/[3H]-cAMP as substrate after 15 mins by liquid scintillation counting analysis | J Med Chem 57: 10304-13 (2014) Article DOI: 10.1021/jm500836h BindingDB Entry DOI: 10.7270/Q28P624W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Phosphodiesterase 7 (Homo sapiens (Human)) | BDBM50034641 (CHEMBL3360415) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Sun Yat-Sen University Curated by ChEMBL | Assay Description Inhibition of PDE7A1 catalytic domain (unknown origin) using [3H]-cGMP/[3H]-cAMP as substrate after 15 mins by liquid scintillation counting analysis | J Med Chem 57: 10304-13 (2014) Article DOI: 10.1021/jm500836h BindingDB Entry DOI: 10.7270/Q28P624W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 3',5'-cyclic phosphodiesterase (Homo sapiens (Human)) | BDBM50034641 (CHEMBL3360415) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
Sun Yat-sen University Curated by ChEMBL | Assay Description Inhibition of PDE9A (unknown origin) | Bioorg Med Chem Lett 30: (2020) | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A (Homo sapiens (Human)) | BDBM50034641 (CHEMBL3360415) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Sun Yat-Sen University Curated by ChEMBL | Assay Description Inhibition of PDE10A2 catalytic domain (unknown origin) using [3H]-cGMP/[3H]-cAMP as substrate after 15 mins by liquid scintillation counting analysi... | J Med Chem 57: 10304-13 (2014) Article DOI: 10.1021/jm500836h BindingDB Entry DOI: 10.7270/Q28P624W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Homo sapiens phosphodiesterase 2A (PDE2A) (Homo sapiens (Human)) | BDBM50034641 (CHEMBL3360415) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Sun Yat-Sen University Curated by ChEMBL | Assay Description Inhibition of PDE2A3 catalytic domain (unknown origin) using [3H]-cGMP/[3H]-cAMP as substrate after 15 mins by liquid scintillation counting analysis | J Med Chem 57: 10304-13 (2014) Article DOI: 10.1021/jm500836h BindingDB Entry DOI: 10.7270/Q28P624W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Phosphodiesterase 3 (PDE3) (Homo sapiens (Human)) | BDBM50034641 (CHEMBL3360415) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.32E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Sun Yat-Sen University Curated by ChEMBL | Assay Description Inhibition of PDE3A catalytic domain (unknown origin) using [3H]-cGMP/[3H]-cAMP as substrate after 15 mins by liquid scintillation counting analysis | J Med Chem 57: 10304-13 (2014) Article DOI: 10.1021/jm500836h BindingDB Entry DOI: 10.7270/Q28P624W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Phosphodiesterase 8 (Homo sapiens (Human)) | BDBM50034641 (CHEMBL3360415) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Sun Yat-Sen University Curated by ChEMBL | Assay Description Inhibition of PDE8A1 catalytic domain (unknown origin) using [3H]-cGMP/[3H]-cAMP as substrate after 15 mins by liquid scintillation counting analysis | J Med Chem 57: 10304-13 (2014) Article DOI: 10.1021/jm500836h BindingDB Entry DOI: 10.7270/Q28P624W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||