 Found 2 hits for monomerid = 50043633
Found 2 hits for monomerid = 50043633 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Serine/threonine-protein kinase WEE1
(Homo sapiens (Human)) | BDBM50043633
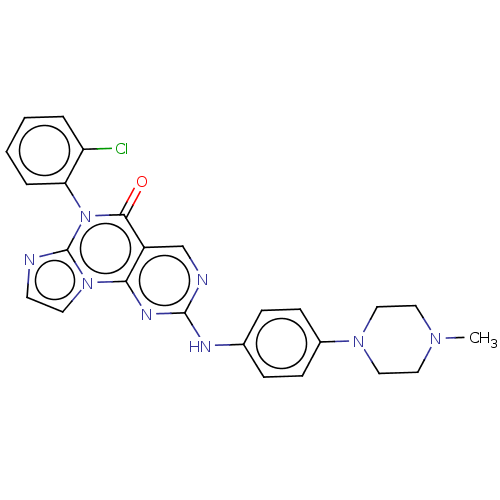
(CHEMBL3355518)Show SMILES CN1CCN(CC1)c1ccc(Nc2ncc3c(n2)n2ccnc2n(-c2ccccc2Cl)c3=O)cc1 |(12.52,-17.19,;12.53,-15.65,;11.2,-14.87,;11.21,-13.33,;12.54,-12.58,;13.88,-13.34,;13.87,-14.88,;12.54,-11.04,;11.21,-10.26,;11.22,-8.72,;12.55,-7.96,;12.56,-6.42,;11.23,-5.65,;11.23,-4.1,;9.9,-3.33,;8.57,-4.09,;8.56,-5.64,;9.89,-6.41,;7.23,-6.4,;6.9,-7.89,;5.39,-8.05,;4.77,-6.65,;5.91,-5.63,;5.91,-4.08,;4.57,-3.32,;3.24,-4.09,;1.9,-3.32,;1.9,-1.77,;3.23,-1,;4.57,-1.77,;5.9,-.99,;7.23,-3.32,;7.23,-1.78,;13.88,-8.72,;13.88,-10.26,)| Show InChI InChI=1S/C25H23ClN8O/c1-31-12-14-32(15-13-31)18-8-6-17(7-9-18)29-24-28-16-19-22(30-24)33-11-10-27-25(33)34(23(19)35)21-5-3-2-4-20(21)26/h2-11,16H,12-15H2,1H3,(H,28,29,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged human Wee1 |
ACS Med Chem Lett 6: 58-62 (2015)
Article DOI: 10.1021/ml5002745
BindingDB Entry DOI: 10.7270/Q2NZ8972 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase WEE1
(Homo sapiens (Human)) | BDBM50043633

(CHEMBL3355518)Show SMILES CN1CCN(CC1)c1ccc(Nc2ncc3c(n2)n2ccnc2n(-c2ccccc2Cl)c3=O)cc1 |(12.52,-17.19,;12.53,-15.65,;11.2,-14.87,;11.21,-13.33,;12.54,-12.58,;13.88,-13.34,;13.87,-14.88,;12.54,-11.04,;11.21,-10.26,;11.22,-8.72,;12.55,-7.96,;12.56,-6.42,;11.23,-5.65,;11.23,-4.1,;9.9,-3.33,;8.57,-4.09,;8.56,-5.64,;9.89,-6.41,;7.23,-6.4,;6.9,-7.89,;5.39,-8.05,;4.77,-6.65,;5.91,-5.63,;5.91,-4.08,;4.57,-3.32,;3.24,-4.09,;1.9,-3.32,;1.9,-1.77,;3.23,-1,;4.57,-1.77,;5.9,-.99,;7.23,-3.32,;7.23,-1.78,;13.88,-8.72,;13.88,-10.26,)| Show InChI InChI=1S/C25H23ClN8O/c1-31-12-14-32(15-13-31)18-8-6-17(7-9-18)29-24-28-16-19-22(30-24)33-11-10-27-25(33)34(23(19)35)21-5-3-2-4-20(21)26/h2-11,16H,12-15H2,1H3,(H,28,29,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 370 | n/a | n/a | n/a | n/a |
AbbVie
Curated by ChEMBL
| Assay Description
Inhibition of human Weel1 in human NCI-H1299 cells assessed as phosphorylation of Cdk1 after overnight incubation by ELISA PD assay |
ACS Med Chem Lett 6: 58-62 (2015)
Article DOI: 10.1021/ml5002745
BindingDB Entry DOI: 10.7270/Q2NZ8972 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data