

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50050964 4-[1-Benzo[1,3]dioxol-5-yl-2-(4-isopropyl-benzenesulfonylamino)-2-oxo-ethoxy]-3-propyl-benzoic acid::CHEMBL8978::L-754142
SMILES: CCCc1cc(ccc1OC(C(=O)NS(=O)(=O)c1ccc(cc1)C(C)C)c1ccc2OCOc2c1)C(O)=O
InChI Key: InChIKey=JPPYWKVKLSBQJM-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Endothelin-1 receptor (Homo sapiens (Human)) | BDBM50050964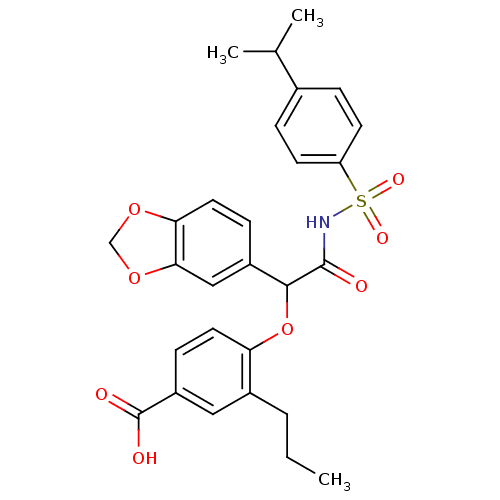 (4-[1-Benzo[1,3]dioxol-5-yl-2-(4-isopropyl-benzenes...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Inhibitory activity of the compound against human Endothelin A receptor in chinese hamster ovary cells | J Med Chem 39: 1039-48 (1996) Article DOI: 10.1021/jm9505369 BindingDB Entry DOI: 10.7270/Q29G5KWT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| EDNRB (Homo sapiens (Human)) | BDBM50050964 (4-[1-Benzo[1,3]dioxol-5-yl-2-(4-isopropyl-benzenes...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 5.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Inhibitory activity of the compound against human Endothelin B receptor in chinese hamster ovary cells | J Med Chem 39: 1039-48 (1996) Article DOI: 10.1021/jm9505369 BindingDB Entry DOI: 10.7270/Q29G5KWT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| EDNRB (Homo sapiens (Human)) | BDBM50050964 (4-[1-Benzo[1,3]dioxol-5-yl-2-(4-isopropyl-benzenes...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Ability of the compound to displace endothelin ([125I]-ET-1) from human Endothelin B receptor | J Med Chem 40: 3217-27 (1997) Article DOI: 10.1021/jm970101g BindingDB Entry DOI: 10.7270/Q2C24VJR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin-1 receptor (Homo sapiens (Human)) | BDBM50050964 (4-[1-Benzo[1,3]dioxol-5-yl-2-(4-isopropyl-benzenes...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.260 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Ability of the compound to displace endothelin ([125I]-ET-1) from human Endothelin A receptor | J Med Chem 40: 3217-27 (1997) Article DOI: 10.1021/jm970101g BindingDB Entry DOI: 10.7270/Q2C24VJR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||