

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50057814 2-{2-[3-(4-Azido-5-hydroxymethyl-tetrahydro-furan-2-yl)-5-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl]-ethyl}-isoindole-1,3-dione::CHEMBL290236
SMILES: Cc1cn(C2CC(N=[N+]=[N-])C(CO)O2)c(=O)n(CCN2C(=O)c3ccccc3C2=O)c1=O
InChI Key: InChIKey=GMWFEPAWHDJWAF-UHFFFAOYSA-N
Data: 1 EC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Human immunodeficiency virus type 1 reverse transcriptase (Human immunodeficiency virus 1) | BDBM50057814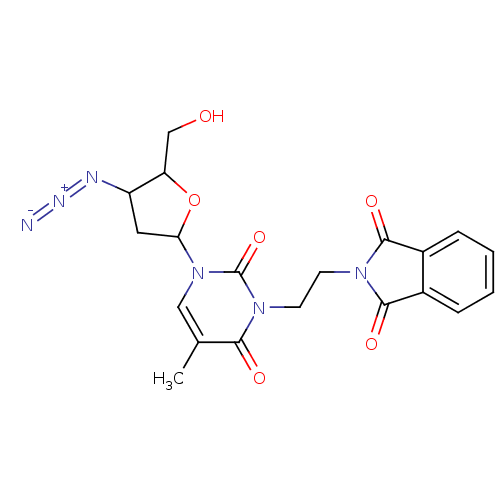 (2-{2-[3-(4-Azido-5-hydroxymethyl-tetrahydro-furan-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.14E+5 | n/a | n/a | n/a | n/a |
Institut de Chimie des Substances Naturelles CNRS Curated by ChEMBL | Assay Description Reverse transcriptase activity was measured in the culture supernatant, concentration that reduces by 50% the HIV produced in the supernatant. | J Med Chem 40: 1550-8 (1997) Article DOI: 10.1021/jm9600095 BindingDB Entry DOI: 10.7270/Q2TH8KSM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||