

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50058817 CHEMBL1271483
SMILES: [H][C@@]12C[C@](C)(CC[C@]1(C)CC[C@]1(C)C2=CC(=O)[C@]2([H])[C@@]3(C)CC[C@H](O)C(C)(C)[C@]3([H])CC[C@@]12C)C(=O)OC
InChI Key: InChIKey=RMIVRCBSQPCSCQ-BDANYOJNSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Carbonic anhydrase (Bos taurus) | BDBM50058817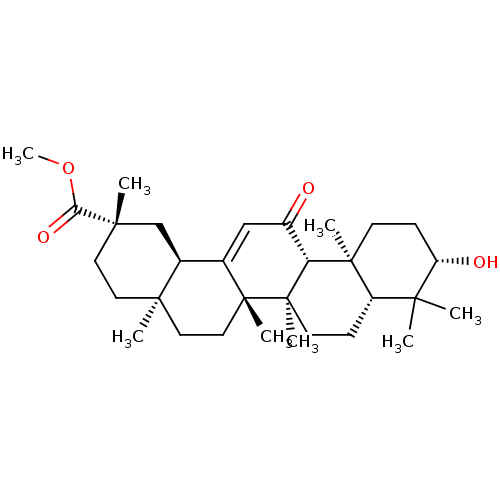 (CHEMBL1271483) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Martin-Luther-Universit£t Halle-Wittenberg Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte carbonic anhydrase 2 using 4-nitrophenyl acetate substrate by photometric assay | Eur J Med Chem 86: 95-102 (2014) Article DOI: 10.1016/j.ejmech.2014.08.051 BindingDB Entry DOI: 10.7270/Q2R78GWB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acyl-CoA: cholesterol acyltransferase (ACAT) (Homo sapiens (Human)) | BDBM50058817 (CHEMBL1271483) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 2.24E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Dalian Institute of Chemical Physics Curated by ChEMBL | Assay Description Inhibition of CE1 in human liver microsomes using 2-(2-Benzoyl-3-methoxyphenyl) benzothiazole as substrate preincubated for 10 mins followed by subst... | Eur J Med Chem 112: 280-8 (2016) BindingDB Entry DOI: 10.7270/Q2TQ63D6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxylesterase 2 (Homo sapiens (Human)) | BDBM50058817 (CHEMBL1271483) | NCI pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 5.61E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Dalian Institute of Chemical Physics Curated by ChEMBL | Assay Description Inhibition of CE2 in human liver microsomes using fluorescein diacetate as substrate preincubated for 10 mins followed by substrate addition measured... | Eur J Med Chem 112: 280-8 (2016) BindingDB Entry DOI: 10.7270/Q2TQ63D6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||