

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50060329 CHEMBL385442::{2-{4-[5-Carboxymethoxy-4-(5-hydroxy-2-methoxy-4-methyl-benzyl)-2-methoxy-benzyl]-5-hydroxy-2-methoxy-benzyl}-5-[4-(2-carboxymethoxy-5-methoxy-benzyl)-2-hydroxy-5-methoxy-benzyl]-4-methoxy-phenoxy}-acetic acid
SMILES: COc1ccc(OCC(O)=O)c(Cc2cc(O)c(Cc3cc(OCC(O)=O)c(Cc4cc(O)c(Cc5cc(OCC(O)=O)c(Cc6cc(O)c(C)cc6OC)cc5OC)cc4OC)cc3OC)cc2OC)c1
InChI Key: InChIKey=QKNYYCRUUWEYEA-UHFFFAOYSA-N
Data: 3 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Leukocyte elastase (Homo sapiens (Human)) | BDBM50060329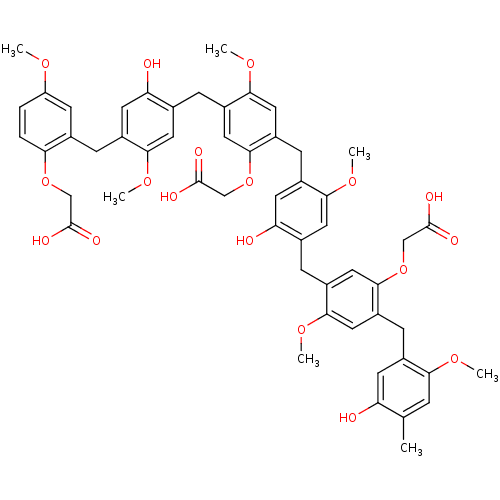 (CHEMBL385442 | {2-{4-[5-Carboxymethoxy-4-(5-hydrox...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibitory activity against human leukocyte elastase | J Med Chem 40: 3408-22 (1997) Article DOI: 10.1021/jm970251r BindingDB Entry DOI: 10.7270/Q26D5S4T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50060329 (CHEMBL385442 | {2-{4-[5-Carboxymethoxy-4-(5-hydrox...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of thrombin at pH 7.5 in HEPES buffer containing 200 mM NaCl with substrate Bz-Phe-Val-Arg-pNA. | J Med Chem 40: 3408-22 (1997) Article DOI: 10.1021/jm970251r BindingDB Entry DOI: 10.7270/Q26D5S4T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cathepsin G (Homo sapiens (Human)) | BDBM50060329 (CHEMBL385442 | {2-{4-[5-Carboxymethoxy-4-(5-hydrox...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of cathepsin G at pH 7.5 in HEPES buffer containing 200 mM NaCl with substrate Suc-Ala-Ala-Pro-Phe-pNA. | J Med Chem 40: 3408-22 (1997) Article DOI: 10.1021/jm970251r BindingDB Entry DOI: 10.7270/Q26D5S4T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||