 Found 12 hits for monomerid = 50060874
Found 12 hits for monomerid = 50060874 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM50060874
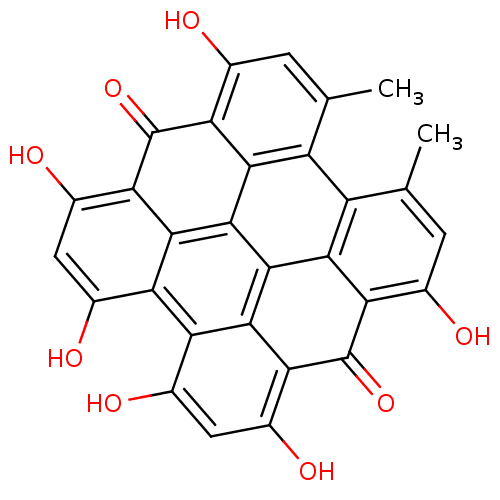
(1,3,4,6,8,13-hexahydroxy-10,11-dimethylphenanthro[...)Show SMILES Cc1cc(O)c2c3c1c1c(C)cc(O)c4c1c1c3c3c(c(O)cc(O)c3c2=O)c2c(O)cc(O)c(c12)c4=O Show InChI InChI=1S/C30H16O8/c1-7-3-9(31)19-23-15(7)16-8(2)4-10(32)20-24(16)28-26-18(12(34)6-14(36)22(26)30(20)38)17-11(33)5-13(35)21(29(19)37)25(17)27(23)28/h3-6,31-36H,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 35 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Westfalische Wilhelms-Universitat
Curated by ChEMBL
| Assay Description
Binding affinity to dopamine D3 receptor |
J Nat Prod 73: 1015-21 (2010)
Article DOI: 10.1021/np1000329
BindingDB Entry DOI: 10.7270/Q2H99642 |
More data for this
Ligand-Target Pair | |
Thioredoxin reductase 1, cytoplasmic
(Rattus norvegicus) | BDBM50060874

(1,3,4,6,8,13-hexahydroxy-10,11-dimethylphenanthro[...)Show SMILES Cc1cc(O)c2c3c1c1c(C)cc(O)c4c1c1c3c3c(c(O)cc(O)c3c2=O)c2c(O)cc(O)c(c12)c4=O Show InChI InChI=1S/C30H16O8/c1-7-3-9(31)19-23-15(7)16-8(2)4-10(32)20-24(16)28-26-18(12(34)6-14(36)22(26)30(20)38)17-11(33)5-13(35)21(29(19)37)25(17)27(23)28/h3-6,31-36H,1-2H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.58E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Padova
Curated by ChEMBL
| Assay Description
Inhibition of rat liver cytosolic TrxR1 by spectrophotometry |
Bioorg Med Chem 19: 631-41 (2011)
Article DOI: 10.1016/j.bmc.2010.10.045
BindingDB Entry DOI: 10.7270/Q25D8S4N |
More data for this
Ligand-Target Pair | |
Thioredoxin reductase 2, mitochondrial
(Rattus norvegicus) | BDBM50060874

(1,3,4,6,8,13-hexahydroxy-10,11-dimethylphenanthro[...)Show SMILES Cc1cc(O)c2c3c1c1c(C)cc(O)c4c1c1c3c3c(c(O)cc(O)c3c2=O)c2c(O)cc(O)c(c12)c4=O Show InChI InChI=1S/C30H16O8/c1-7-3-9(31)19-23-15(7)16-8(2)4-10(32)20-24(16)28-26-18(12(34)6-14(36)22(26)30(20)38)17-11(33)5-13(35)21(29(19)37)25(17)27(23)28/h3-6,31-36H,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.32E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Padova
Curated by ChEMBL
| Assay Description
Inhibition of rat liver mitochondrial TrxR2 by spectrophotometry |
Bioorg Med Chem 19: 631-41 (2011)
Article DOI: 10.1016/j.bmc.2010.10.045
BindingDB Entry DOI: 10.7270/Q25D8S4N |
More data for this
Ligand-Target Pair | |
Corticotropin releasing factor receptor 1
(Homo sapiens (Human)) | BDBM50060874

(1,3,4,6,8,13-hexahydroxy-10,11-dimethylphenanthro[...)Show SMILES Cc1cc(O)c2c3c1c1c(C)cc(O)c4c1c1c3c3c(c(O)cc(O)c3c2=O)c2c(O)cc(O)c(c12)c4=O Show InChI InChI=1S/C30H16O8/c1-7-3-9(31)19-23-15(7)16-8(2)4-10(32)20-24(16)28-26-18(12(34)6-14(36)22(26)30(20)38)17-11(33)5-13(35)21(29(19)37)25(17)27(23)28/h3-6,31-36H,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Westfalische Wilhelms-Universitat
Curated by ChEMBL
| Assay Description
Inhibition of CRF1 receptor |
J Nat Prod 73: 1015-21 (2010)
Article DOI: 10.1021/np1000329
BindingDB Entry DOI: 10.7270/Q2H99642 |
More data for this
Ligand-Target Pair | |
Thioredoxin reductase 1, cytoplasmic
(Rattus norvegicus) | BDBM50060874

(1,3,4,6,8,13-hexahydroxy-10,11-dimethylphenanthro[...)Show SMILES Cc1cc(O)c2c3c1c1c(C)cc(O)c4c1c1c3c3c(c(O)cc(O)c3c2=O)c2c(O)cc(O)c(c12)c4=O Show InChI InChI=1S/C30H16O8/c1-7-3-9(31)19-23-15(7)16-8(2)4-10(32)20-24(16)28-26-18(12(34)6-14(36)22(26)30(20)38)17-11(33)5-13(35)21(29(19)37)25(17)27(23)28/h3-6,31-36H,1-2H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.58E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Padova
Curated by ChEMBL
| Assay Description
Inhibition of rat liver cytosolic TrxR1 by spectrophotometry |
Bioorg Med Chem 19: 631-41 (2011)
Article DOI: 10.1016/j.bmc.2010.10.045
BindingDB Entry DOI: 10.7270/Q25D8S4N |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50060874

(1,3,4,6,8,13-hexahydroxy-10,11-dimethylphenanthro[...)Show SMILES Cc1cc(O)c2c3c1c1c(C)cc(O)c4c1c1c3c3c(c(O)cc(O)c3c2=O)c2c(O)cc(O)c(c12)c4=O Show InChI InChI=1S/C30H16O8/c1-7-3-9(31)19-23-15(7)16-8(2)4-10(32)20-24(16)28-26-18(12(34)6-14(36)22(26)30(20)38)17-11(33)5-13(35)21(29(19)37)25(17)27(23)28/h3-6,31-36H,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Westfalische Wilhelms-Universitat
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 |
J Nat Prod 73: 1015-21 (2010)
Article DOI: 10.1021/np1000329
BindingDB Entry DOI: 10.7270/Q2H99642 |
More data for this
Ligand-Target Pair | |
Glutathione reductase
(Saccharomyces cerevisiae) | BDBM50060874

(1,3,4,6,8,13-hexahydroxy-10,11-dimethylphenanthro[...)Show SMILES Cc1cc(O)c2c3c1c1c(C)cc(O)c4c1c1c3c3c(c(O)cc(O)c3c2=O)c2c(O)cc(O)c(c12)c4=O Show InChI InChI=1S/C30H16O8/c1-7-3-9(31)19-23-15(7)16-8(2)4-10(32)20-24(16)28-26-18(12(34)6-14(36)22(26)30(20)38)17-11(33)5-13(35)21(29(19)37)25(17)27(23)28/h3-6,31-36H,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.67E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Padova
Curated by ChEMBL
| Assay Description
Inhibition of yeast glutathione reductase by spectrophotometry |
Bioorg Med Chem 19: 631-41 (2011)
Article DOI: 10.1016/j.bmc.2010.10.045
BindingDB Entry DOI: 10.7270/Q25D8S4N |
More data for this
Ligand-Target Pair | |
Human immunodeficiency virus type 1 integrase
(Human immunodeficiency virus 1) | BDBM50060874

(1,3,4,6,8,13-hexahydroxy-10,11-dimethylphenanthro[...)Show SMILES Cc1cc(O)c2c3c1c1c(C)cc(O)c4c1c1c3c3c(c(O)cc(O)c3c2=O)c2c(O)cc(O)c(c12)c4=O Show InChI InChI=1S/C30H16O8/c1-7-3-9(31)19-23-15(7)16-8(2)4-10(32)20-24(16)28-26-18(12(34)6-14(36)22(26)30(20)38)17-11(33)5-13(35)21(29(19)37)25(17)27(23)28/h3-6,31-36H,1-2H3 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of California
Curated by ChEMBL
| Assay Description
Inhibition of HIV-1 integrase, under 1 uM for the 3''-preprocessing |
J Med Chem 43: 2100-14 (2000)
BindingDB Entry DOI: 10.7270/Q27D2VTS |
More data for this
Ligand-Target Pair | |
cysteine protease ATG4B isoform a
(Homo sapiens (Human)) | BDBM50060874

(1,3,4,6,8,13-hexahydroxy-10,11-dimethylphenanthro[...)Show SMILES Cc1cc(O)c2c3c1c1c(C)cc(O)c4c1c1c3c3c(c(O)cc(O)c3c2=O)c2c(O)cc(O)c(c12)c4=O Show InChI InChI=1S/C30H16O8/c1-7-3-9(31)19-23-15(7)16-8(2)4-10(32)20-24(16)28-26-18(12(34)6-14(36)22(26)30(20)38)17-11(33)5-13(35)21(29(19)37)25(17)27(23)28/h3-6,31-36H,1-2H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of Atg4B (unknown origin) using YFP-LC3B-EmGFP as substrate after 40 mins by FRET-based assay |
Eur J Med Chem 123: 631-638 (2016)
Article DOI: 10.1016/j.ejmech.2016.07.073
BindingDB Entry DOI: 10.7270/Q2FT8P1P |
More data for this
Ligand-Target Pair | |
Corticotropin releasing factor receptor 1
(Homo sapiens (Human)) | BDBM50060874

(1,3,4,6,8,13-hexahydroxy-10,11-dimethylphenanthro[...)Show SMILES Cc1cc(O)c2c3c1c1c(C)cc(O)c4c1c1c3c3c(c(O)cc(O)c3c2=O)c2c(O)cc(O)c(c12)c4=O Show InChI InChI=1S/C30H16O8/c1-7-3-9(31)19-23-15(7)16-8(2)4-10(32)20-24(16)28-26-18(12(34)6-14(36)22(26)30(20)38)17-11(33)5-13(35)21(29(19)37)25(17)27(23)28/h3-6,31-36H,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Westfalische Wilhelms-Universitat
Curated by ChEMBL
| Assay Description
Inhibition of recombinant CRF1 receptor expressed in CHO cells assessed as inhibition of CRF-induced cAMP formation |
J Nat Prod 73: 1015-21 (2010)
Article DOI: 10.1021/np1000329
BindingDB Entry DOI: 10.7270/Q2H99642 |
More data for this
Ligand-Target Pair | |
Arachidonate 5-lipoxygenase
(Bos taurus) | BDBM50060874

(1,3,4,6,8,13-hexahydroxy-10,11-dimethylphenanthro[...)Show SMILES Cc1cc(O)c2c3c1c1c(C)cc(O)c4c1c1c3c3c(c(O)cc(O)c3c2=O)c2c(O)cc(O)c(c12)c4=O Show InChI InChI=1S/C30H16O8/c1-7-3-9(31)19-23-15(7)16-8(2)4-10(32)20-24(16)28-26-18(12(34)6-14(36)22(26)30(20)38)17-11(33)5-13(35)21(29(19)37)25(17)27(23)28/h3-6,31-36H,1-2H3 | PDB
MMDB
Reactome pathway
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Westfälische Wilhelms-Universität Münster
Curated by ChEMBL
| Assay Description
5-LO inhibitory activity was determined by inhibition of LTB4 biosynthesis in bovine polymorphonuclear leukocytes (PMNL) |
J Med Chem 40: 3773-80 (1997)
Article DOI: 10.1021/jm970292n
BindingDB Entry DOI: 10.7270/Q20R9NJN |
More data for this
Ligand-Target Pair | |
Thioredoxin reductase 2, mitochondrial
(Rattus norvegicus) | BDBM50060874

(1,3,4,6,8,13-hexahydroxy-10,11-dimethylphenanthro[...)Show SMILES Cc1cc(O)c2c3c1c1c(C)cc(O)c4c1c1c3c3c(c(O)cc(O)c3c2=O)c2c(O)cc(O)c(c12)c4=O Show InChI InChI=1S/C30H16O8/c1-7-3-9(31)19-23-15(7)16-8(2)4-10(32)20-24(16)28-26-18(12(34)6-14(36)22(26)30(20)38)17-11(33)5-13(35)21(29(19)37)25(17)27(23)28/h3-6,31-36H,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.32E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Padova
Curated by ChEMBL
| Assay Description
Inhibition of rat liver mitochondrial TrxR2 by spectrophotometry |
Bioorg Med Chem 19: 631-41 (2011)
Article DOI: 10.1016/j.bmc.2010.10.045
BindingDB Entry DOI: 10.7270/Q25D8S4N |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data