

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50065304 3-[1-(4-Hydroxy-phenyl)-meth-(Z)-ylidene]-1,3-dihydro-indol-2-one::CHEMBL313578
SMILES: Oc1ccc(\C=C2/C(=O)Nc3ccccc23)cc1
InChI Key: InChIKey=PLAOAGFXNCEZMZ-LCYFTJDESA-N
Data: 7 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Serine/threonine-protein kinase PLK1 (Homo sapiens (Human)) | BDBM50065304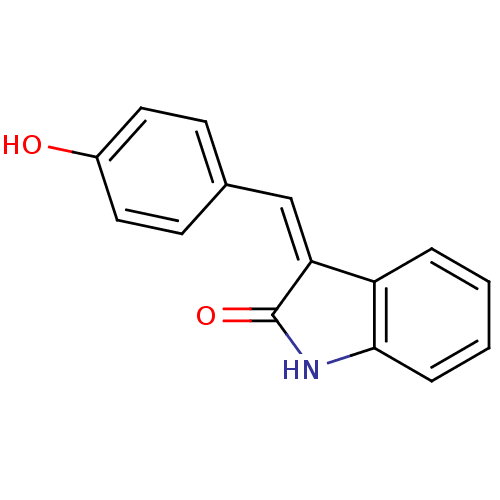 (3-[1-(4-Hydroxy-phenyl)-meth-(Z)-ylidene]-1,3-dihy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
EntreMed Inc. Curated by ChEMBL | Assay Description Inhibition of PLK1 (unknown origin) by FRET-based homogeneous assay | J Med Chem 56: 6069-87 (2013) Article DOI: 10.1021/jm400380m BindingDB Entry DOI: 10.7270/Q2HM59V9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine/threonine-protein kinase PLK4 (Homo sapiens (Human)) | BDBM50065304 (3-[1-(4-Hydroxy-phenyl)-meth-(Z)-ylidene]-1,3-dihy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 230 | n/a | n/a | n/a | n/a | n/a | n/a |
EntreMed Inc. Curated by ChEMBL | Assay Description Inhibition of N-terminal GST-tagged human PLK4 (1 to 391 amino acids) expressed in Escherichia coli using TMB as substrate after 30 mins by indirect ... | J Med Chem 56: 6069-87 (2013) Article DOI: 10.1021/jm400380m BindingDB Entry DOI: 10.7270/Q2HM59V9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Receptor tyrosine-protein kinase erbB-2 (Homo sapiens (Human)) | BDBM50065304 (3-[1-(4-Hydroxy-phenyl)-meth-(Z)-ylidene]-1,3-dihy...) | PDB MMDB UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
SUGEN, Inc. Curated by ChEMBL | Assay Description Concentration required to achieve 50% inhibition of tyrosine phosphorylation on human Her-2 receptor tyrosine kinase (HER-2 RTK) | J Med Chem 41: 2588-603 (1998) Article DOI: 10.1021/jm980123i BindingDB Entry DOI: 10.7270/Q2G73CTT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin-like growth factor I receptor (Homo sapiens (Human)) | BDBM50065304 (3-[1-(4-Hydroxy-phenyl)-meth-(Z)-ylidene]-1,3-dihy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
SUGEN, Inc. Curated by ChEMBL | Assay Description Concentration required to achieve 50% inhibition of tyrosine phosphorylation on human Insulin-like growth factor I receptor | J Med Chem 41: 2588-603 (1998) Article DOI: 10.1021/jm980123i BindingDB Entry DOI: 10.7270/Q2G73CTT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vascular endothelial growth factor receptor 1 (Mus musculus) | BDBM50065304 (3-[1-(4-Hydroxy-phenyl)-meth-(Z)-ylidene]-1,3-dihy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
SUGEN, Inc. Curated by ChEMBL | Assay Description Concentration required to achieve 50% inhibition of tyrosine phosphorylation on murine VEGF receptor (FLK-1 RTK). | J Med Chem 41: 2588-603 (1998) Article DOI: 10.1021/jm980123i BindingDB Entry DOI: 10.7270/Q2G73CTT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50065304 (3-[1-(4-Hydroxy-phenyl)-meth-(Z)-ylidene]-1,3-dihy...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
SUGEN, Inc. Curated by ChEMBL | Assay Description Concentration required to achieve 50% inhibition of tyrosine phosphorylation on human Epidermal growth factor receptor (EGF RTK). | J Med Chem 41: 2588-603 (1998) Article DOI: 10.1021/jm980123i BindingDB Entry DOI: 10.7270/Q2G73CTT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Platelet-derived growth factor receptor beta (Homo sapiens (Human)) | BDBM50065304 (3-[1-(4-Hydroxy-phenyl)-meth-(Z)-ylidene]-1,3-dihy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 8.39E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
SUGEN, Inc. Curated by ChEMBL | Assay Description Test concentration required to achieve 50% inhibition of tyrosine phosphorylation on human Platelet-derived growth factor receptor beta (PDGF RTK). | J Med Chem 41: 2588-603 (1998) Article DOI: 10.1021/jm980123i BindingDB Entry DOI: 10.7270/Q2G73CTT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||