

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50073429 CHEMBL82032::CHEMBL84575::N-((S)-1-Carbamimidoyl-2-hydroxy-piperidin-3-yl)-2-((S)-2-oxo-3-phenylmethanesulfonylamino-azepan-1-yl)-acetamide
SMILES: NC(=N)N1CCC[C@H](NC(=O)CN2CCCC[C@H](NS(=O)(=O)Cc3ccccc3)C2=O)C1O
InChI Key: InChIKey=PIIUKKBKYNGKMK-NBJLRHFZSA-N
Data: 10 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Trypsin-1 (Homo sapiens (Human)) | BDBM50073429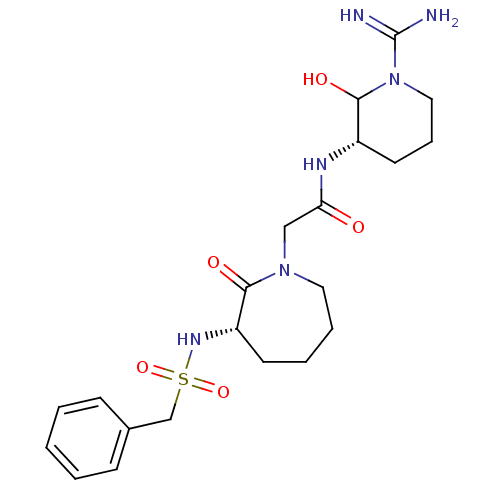 (CHEMBL82032 | CHEMBL84575 | N-((S)-1-Carbamimidoyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 101 | n/a | n/a | n/a | n/a | n/a | n/a |
Corvas International, Inc. Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound was evaluated against human trypsin | Bioorg Med Chem Lett 8: 3683-8 (1999) BindingDB Entry DOI: 10.7270/Q2ZP459T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasmin (Rattus norvegicus) | BDBM50073429 (CHEMBL82032 | CHEMBL84575 | N-((S)-1-Carbamimidoyl...) | MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | >2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Corvas International, Inc. Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound was evaluated against thrombolytic enzyme plasmin | Bioorg Med Chem Lett 8: 3683-8 (1999) BindingDB Entry DOI: 10.7270/Q2ZP459T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50073429 (CHEMBL82032 | CHEMBL84575 | N-((S)-1-Carbamimidoyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Corvas International, Inc. Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound was evaluated against Coagulation factor X | Bioorg Med Chem Lett 8: 3683-8 (1999) BindingDB Entry DOI: 10.7270/Q2ZP459T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50073429 (CHEMBL82032 | CHEMBL84575 | N-((S)-1-Carbamimidoyl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.710 | n/a | n/a | n/a | n/a | n/a | n/a |
Corvas International, Inc. Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against human enzyme thrombin | Bioorg Med Chem Lett 9: 2573-8 (1999) BindingDB Entry DOI: 10.7270/Q26M361R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50073429 (CHEMBL82032 | CHEMBL84575 | N-((S)-1-Carbamimidoyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description The compound was tested in vitro for inhibitory activity against Coagulation factor X | Bioorg Med Chem Lett 7: 2421-2426 (1997) Article DOI: 10.1016/S0960-894X(97)00446-0 BindingDB Entry DOI: 10.7270/Q2X92B91 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Trypsin-1 (Homo sapiens (Human)) | BDBM50073429 (CHEMBL82032 | CHEMBL84575 | N-((S)-1-Carbamimidoyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 75 | n/a | n/a | n/a | n/a | n/a | n/a |
Corvas International, Inc. Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against human enzyme trypsin | Bioorg Med Chem Lett 9: 2573-8 (1999) BindingDB Entry DOI: 10.7270/Q26M361R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasminogen (Homo sapiens (Human)) | BDBM50073429 (CHEMBL82032 | CHEMBL84575 | N-((S)-1-Carbamimidoyl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | >2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Corvas International, Inc. Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against human enzyme plasmin | Bioorg Med Chem Lett 9: 2573-8 (1999) BindingDB Entry DOI: 10.7270/Q26M361R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50073429 (CHEMBL82032 | CHEMBL84575 | N-((S)-1-Carbamimidoyl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | n/a | n/a | 0.710 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description The compound was tested in vitro for inhibitory activity against Coagulation factor II | Bioorg Med Chem Lett 7: 2421-2426 (1997) Article DOI: 10.1016/S0960-894X(97)00446-0 BindingDB Entry DOI: 10.7270/Q2X92B91 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Trypsin-1 (Homo sapiens (Human)) | BDBM50073429 (CHEMBL82032 | CHEMBL84575 | N-((S)-1-Carbamimidoyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | n/a | n/a | 152 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description The compound was tested in vitro for inhibitory activity against human trypsin | Bioorg Med Chem Lett 7: 2421-2426 (1997) Article DOI: 10.1016/S0960-894X(97)00446-0 BindingDB Entry DOI: 10.7270/Q2X92B91 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50073429 (CHEMBL82032 | CHEMBL84575 | N-((S)-1-Carbamimidoyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Corvas International, Inc. Curated by ChEMBL | Assay Description In vitro inhibitory activity of the compound against human enzyme Coagulation factor X | Bioorg Med Chem Lett 9: 2573-8 (1999) BindingDB Entry DOI: 10.7270/Q26M361R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||